Hydroxylammonium sulfate
 | |
| Names | |
|---|---|
| Other names
Hydroxylamine sulfate
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.030.095 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 2865 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
H8N2O6S |
| Molar mass | 164.14 g/mol |
| Appearance | white crystalline to fine product, slightly hygroscopic |
| Density | 1.88 g/cm3 |
| Melting point | 120 °C (248 °F; 393 K) decomposes |
Solubility in water
|
58.7 g/100 ml (20 °C) |
| Structure[1] | |
Crystal structure
|
Monoclinic |
Space group
|
P21/c |
Lattice constant
|
a = 7.932±0.002 Å, b = 7.321±0.002 Å, c = 10.403±0.003 Å α = 90°, β = 106.93±0.03°, γ = 90°
|
Formula units (Z)
|
4 |
| Hazards | |
| GHS labelling: | |
Pictograms
|
   
|
Signal word
|
Warning |
Hazard statements
|
H290, H302, H312, H315, H317, H319, H351, H373, H400, H412 |
Precautionary statements
|
P201, P202, P234, P260, P264, P270, P272, P273, P280, P281, P301+P312, P302+P352, P305+P351+P338, P308+P313, P312, P314, P321, P322, P330, P332+P313, P333+P313, P337+P313, P362, P363, P390, P391, P404, P405, P501 |
| NFPA 704 (fire diamond) | 
3
1
2 |
| Safety data sheet (SDS) | External MSDS |
| Related compounds | |
Other anions
|
Hydroxylammonium nitrate Hydroxylammonium chloride |
Other cations
|
Ammonium sulfate Hydrazinium sulfate |
Related compounds
|
Hydroxylamine |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
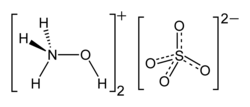
Hydroxylammonium sulfate is the inorganic compound with the formula [NH3OH]2SO4. A colorless solid, it is the sulfate salt of hydroxylamine. It is primarily used as an easily handled form of hydroxylamine, which is a volatile liquid.[2]
Production
Hydroxylammonium sulfate is prepared industrially by protonation of hydroxylamine. The latter is produced by the hydrogenation of nitric oxide using a platinum catalyst:[2]
- 2 NO + 3 H2 + H2SO4 → [NH3OH]2[SO4]
Another route to NH2OH is the Raschig process: aqueous ammonium nitrite is reduced by HSO−3 and SO2 at 0 °C to yield a hydroxylamido-N,N-disulfonate anion:[3]
- [NH4]+[NO2]− + 2 SO2 + NH3 + H2O → [NH4]2[HON(SO3)2]
This ammonium hydroxylamine disulfonate anion is then hydrolyzed to give hydroxylammonium sulfate:
- [NH4]2[HON(SO3)2] + 2 H2O → [HONH3]2SO4
Applications
Almost all hydroxylamine and its salts are used to make precursors to nylons via cyclohexanone oxime.[2] Many aldehydes and ketones undergo the same conversion to oximes. carboxylic acids and their derivatives (e.g. esters) convert to hydroxamic acids. Isocyanates to N-hydroxyureas. Nitriles react to give amidoximes. Hydroxylammonium sulfate is also used to generate hydroxylamine-O-sulfonic acid from oleum or from chlorosulfuric acid.
Hydroxylammonium sulfate is used in the production of anti-skinning agents, pharmaceuticals, rubber, textiles, plastics and detergents. It is a radical scavenger that terminates radical polymerization reactions and serves as an antioxidant in natural rubber. (NH3OH)2SO4 is a starting material for some insecticides, herbicides and growth regulators. It is used in photography as a stabiliser for colour developers and as an additive in photographic emulsions in colour film.
Structure
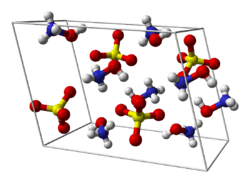
Hydroxylammonium sulfate exists as tetrahedral NH3OH+ cations and sulfate anions.
Safety
Hydroxylamine, which occurs widely in nature, has low toxicity. The compound is stable below 120 °C.[2]
References
- ^ Mirceva, A.; Golic, L. (15 May 1995). "Hydroxylammonium Sulfate". Acta Crystallographica Section C Crystal Structure Communications. 51 (5): 798–800. Bibcode:1995AcCrC..51..798M. doi:10.1107/S0108270194013351.
- ^ a b c d Ritz, Josef; Fuchs, Hugo; Perryman, Howard G. (2000). "Hydroxylamine". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a13_527. ISBN 3527306730.
- ^ Oblath, S. B.; Markowitz, S. S.; Novakov, T.; Chang, S. G. (December 1982). "Kinetics of the initial reaction of nitrite ion in bisulfite solutions" (PDF). The Journal of Physical Chemistry. 86 (25): 4853–4857. doi:10.1021/j100222a005. OSTI 1149469.