Dextrothyroxine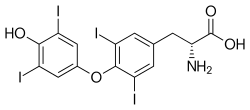 |
|
| Trade names | Choloxin |
|---|
| Other names | D-3,5,3',5'-tetraiodothyronine |
|---|
| AHFS/Drugs.com | Multum Consumer Information |
|---|
| ATC code | |
|---|
|
(2R)-2-amino-3-[4-(4-hydroxy-3,5-diiodophenoxy)-3,5-diiodophenyl]propanoic acid
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| ChEBI | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
| ECHA InfoCard | 100.000.094 |
|---|
|
| Formula | C15H11I4NO4 |
|---|
| Molar mass | 776.874 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
O=C(O)[C@H](N)Cc2cc(I)c(Oc1cc(I)c(O)c(I)c1)c(I)c2
|
InChI=1S/C15H11I4NO4/c16-8-4-7(5-9(17)13(8)21)24-14-10(18)1-6(2-11(14)19)3-12(20)15(22)23/h1-2,4-5,12,21H,3,20H2,(H,22,23)/t12-/m1/s1 Key:XUIIKFGFIJCVMT-GFCCVEGCSA-N
|
Dextrothyroxine (trade name Choloxin) is a dextrorotary isomer of thyroxine.[1] It saw research as a cholesterol-lowering drug[2] but was pulled due to cardiac side-effects. It increases hepatic lipase which in turn improves utilization of triglycerides and decreases levels of lipoprotein(a) in blood serum.[3]
References
- ^ "Dextrothyroxine (Code C61719)". NCI Thesaurus. National Cancer Institute. 2011-11-14. Retrieved 2020-01-28.
- ^ Bantle JP, Hunninghake DB, Frantz ID, Kuba K, Mariash CN, Oppenheimer JH (September 1984). "Comparison of effectiveness of thyrotropin-suppressive doses of D- and L-thyroxine in treatment of hypercholesterolemia". The American Journal of Medicine. 77 (3): 475–81. doi:10.1016/0002-9343(84)90107-4. PMID 6475988.
- ^ Bommer C, Werle E, Walter-Sack I, Keller C, Gehlen F, Wanner C, et al. (January 1998). "D-thyroxine reduces lipoprotein(a) serum concentration in dialysis patients". Journal of the American Society of Nephrology. 9 (1): 90–6. doi:10.1681/ASN.V9190. PMID 9440092.
Lipid-lowering agents (C10) |
|---|
| GI tract | | Cholesterol absorption inhibitors, NPC1L1 | |
|---|
| Bile acid sequestrants/resins (LDL) | |
|---|
|
|---|
| Liver | | Statins (HMG-CoA reductase, LDL) | |
|---|
| Niacin and derivatives (HDL and LDL) | |
|---|
| MTTP inhibitors (VLDL) |
- Dirlotapide
- Lomitapide
- Mitratapide
|
|---|
| ATP citrate lyase inhibitors (LDL) | |
|---|
| Thyromimetics (VLDL) | |
|---|
|
|---|
| Blood vessels | | PPAR agonists (LDL) | |
|---|
| CETP inhibitors (HDL) |
- Anacetrapib†
- Dalcetrapib§
- Evacetrapib§
- Obicetrapib†
- Torcetrapib§
|
|---|
| PCSK9 inhibitors (LDL) |
- Alirocumab
- Bococizumab
- Enlicitide†
- Evolocumab
- Inclisiran
- Lerodalcibep
|
|---|
| ANGPTL3 inhibitors (LDL/HDL) | |
|---|
|
|---|
| Combinations |
- Amlodipine/atorvastatin
- Bempedoic acid/ezetimibe
- Ezetimibe/atorvastatin
- Ezetimibe/rosuvastatin
- Ezetimibe/simvastatin
- Fenofibrate/pravastatin
- Fenofibrate/simvastatin
- Niacin/laropiprant
- Niacin/lovastatin
- Niacin/simvastatin
|
|---|
| Other |
- Alipogene tiparvovec
- Azacosterol
- Azalanstat
- Benfluorex‡
- Darapladib§
- Lapaquistat§
- Magnesium pyridoxal 5-phosphate glutamate
- Meglutol
- Mipomersen
- Olezarsen
- Omega−3-acid ethyl esters
- Plozasiran
- Policosanol
- Probucol
- Tiadenol
- Triparanol‡
- Volanesorsen
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
Thyroid hormone receptor modulators |
|---|
Receptor
(ligands) | | THRTooltip Thyroid hormone receptor | | Agonists | |
|---|
Thyromimetics
(selective agonists) |
- Elunetirom (ABX-002, MA-JD21)
- Eprotirome (KB-2115)
- KB-141
- KB-2611
- KB-130015
- MB-07344
- MB-07811
- Resmetirom
- Sobetirome (GC-1, GRX-431)
- VK-0214
- VK-2809
- ZYT1
|
|---|
| Antagonists |
- 1-850
- NH3
- Tetraiodothyroacetic acid (Tetrac)
|
|---|
|
|---|
|
|---|
Transporter
(blockers) | NISTooltip Sodium-iodide symporter
|
- Inhibitors: Cyanogenic glycosides
- Perchlorates (e.g., potassium perchlorate)
- Pertechnetates (e.g., sodium pertechnetate)
- Thiocyanates
|
|---|
|
|---|
Enzyme
(inhibitors) | | TPOTooltip Thyroid peroxidase | |
|---|
| DIOTooltip Iodothyronine deiodinase | |
|---|
|
|---|
| Others | |
|---|
- See also: Receptor/signaling modulators
|
