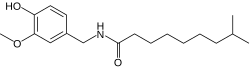
Dihydrocapsaicin
Names
Preferred IUPAC name
N -[(4-Hydroxy-3-methoxyphenyl)methyl]-8-methylnonanamide
Other names
Dihydrocapsaicin, 6,7-Dihydrocapsaicin, 8-Methyl-N -vanillylnonanamide, Vanillylamide of 8-methylnonanoic acid, DHC, CCRIS 1589
Identifiers
CAS Number
3D model (JSmol)
Beilstein Reference
2815150
ChEBI
ChEMBL
ChemSpider
ECHA InfoCard
100.115.366
EC Number
KEGG
RTECS number
UNII
InChI=1S/C18H29NO3/c1-14(2)8-6-4-5-7-9-18(21)19-13-15-10-11-16(20)17(12-15)22-3/h10-12,14,20H,4-9,13H2,1-3H3,(H,19,21)
N Key: XJQPQKLURWNAAH-UHFFFAOYSA-N
N InChI=1/C18H29NO3/c1-14(2)8-6-4-5-7-9-18(21)19-13-15-10-11-16(20)17(12-15)22-3/h10-12,14,20H,4-9,13H2,1-3H3,(H,19,21)
Key: XJQPQKLURWNAAH-UHFFFAOYAI
CC(C)CCCCCCC(=O)NCC1=CC(=C(C=C1)O)OC
Properties
Chemical formula
C18 H29 NO3
Molar mass
307.43 g/mol
Appearance
White to off-white solid
Solubility in water
Sparingly soluble
Hazards
GHS labelling:
Pictograms
Signal word
Danger
Hazard statements
H301 , H315 , H319 , H335
Precautionary statements
P261 , P264 , P270 , P271 , P280 , P301+P310 , P302+P352 , P304+P340 , P305+P351+P338 , P312 , P321 , P330 , P332+P313 , P337+P313 , P362 , P403+P233 , P405 , P501
NFPA 704 (fire diamond)
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Dihydrocapsaicin Heat Above peaktoxic ) Scoville scale 16,000,000[ 1]
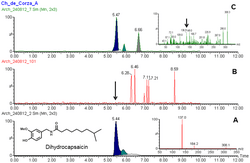
MS/MS spectra of standard dihydrocapsaicin (A) and from sample extract (B). Sample B confirms the compound was found in prehispanic pottery from Mexico. See here for details doi:10.1371/journal.pone.0079013.g005 Dihydrocapsaicin is a capsaicinoid and analog and congener of capsaicin in chili peppers (Capsicum ). Like capsaicin, it is an irritant. It accounts for about 22% of the total capsaicinoid mixture[ 2] [ 1] dimethyl sulfoxide and 100% ethanol .
See also
References
^ a b Govindarajan, Sathyanarayana (1991). "Capsicum — Production, Technology, Chemistry, and Quality. Part V. Impact on Physiology, Pharmacology, Nutrition, and Metabolism; Structure, Pungency, Pain, and Desensitization Sequences". Critical Reviews in Food Science and Nutrition . 29 (6): 435– 474. doi:10.1080/10408399109527536 . PMID 2039598 . ^ Bennett DJ, Kirby GW (1968). "Constitution and biosynthesis of capsaicin". J. Chem. Soc. C : 442. doi:10.1039/j39680000442 .
External links
TRPA
Activators
4-Hydroxynonenal 4-Oxo-2-nonenal
5,6-EET
12S-HpETE
15-Deoxy-Δ12,14 -prostaglandin J2
α-Sanshool (ginger , Sichuan and melegueta peppers)
Acrolein Allicin (garlic)Allyl isothiocyanate (mustard, radish, horseradish, wasabi)AM404
ASP-7663
Bradykinin Cannabichromene (cannabis)Cannabidiol (cannabis)Cannabigerol (cannabis)
Cinnamaldehyde (cinnamon)CR gas (dibenzoxazepine; DBO)
CS gas (2-chlorobenzal malononitrile) Cuminaldehyde (cumin)Curcumin (turmeric)Dehydroligustilide (celery)
Diallyl disulfide Dicentrine (Lindera spp.)
Farnesyl thiosalicylic acid
Formalin Gingerols (ginger )Hepoxilin A3
Hepoxilin B3
Hydrogen peroxide Icilin
Isothiocyanate
JT-010
Ligustilide (celery, Angelica acutiloba )
Linalool (Sichuan pepper, thyme)Methylglyoxal Methyl salicylate (wintergreen)N-Methylmaleimide Nicotine (tobacco)Oleocanthal (olive oil)
Paclitaxel (Pacific yew)PF-4840154
Phenacyl chloride Polygodial (Dorrigo pepper)Shogaols (ginger , Sichuan and melegueta peppers)
Tear gases
Tetrahydrocannabinol (cannabis)Tetrahydrocannabiorcol
Thiopropanal S-oxide (onion)Umbellulone (Umbellularia californica )
WIN 55,212-2 Blockers
A-967079
AM-0902
Dehydroligustilide (celery)
HC-030031
Nicotine (tobacco)PF-04745637
Ruthenium red
TRPC
Activators
Adhyperforin (St John's wort)
Diacyl glycerol
GSK1702934A
Hyperforin (St John's wort)Substance P Blockers
TRPM
TRPML
Activators
EVP21
MK6-83
ML-SA1
ML2-SA1
PI(3,5)P2
SF-22
SN-2 Blockers
TRPP
Activators
Triptolide (Tripterygium wilfordii ) Blockers
TRPV
Activators
2-APB
5,6-EET
9-HODE
9-oxoODE
12S-HETE
12S-HpETE
13-HODE
13-oxoODE
20-HETE
α-Sanshool (ginger , Sichuan and melegueta peppers)
Allicin (garlic)AM404
Anandamide Bisandrographolide (Andrographis paniculata )
Camphor (camphor laurel, rosemary, camphorweed, African blue basil, camphor basil)Cannabidiol (cannabis)Cannabidivarin (cannabis)
Capsaicin (chili pepper)Carvacrol (oregano, thyme, pepperwort, wild bergamot, others)DHEA Diacyl glycerol
Dihydrocapsaicin (chili pepper)Estradiol Eugenol (basil, clove)Evodiamine (Euodia ruticarpa )Gingerols (ginger )GSK1016790A
Heat
Hepoxilin A3
Hepoxilin B3
Homocapsaicin (chili pepper)Homodihydrocapsaicin (chili pepper)Incensole (incense)
Lysophosphatidic acid Low pH (acidic conditions)
Menthol (mint)N-Arachidonoyl dopamine
N-Oleoyldopamine
N-Oleoylethanolamide
Nonivamide (PAVA) (PAVA spray)
Nordihydrocapsaicin (chili pepper)
Paclitaxel (Pacific yew)Paracetamol (acetaminophen) Phenylacetylrinvanil
Phorbol esters (e.g., 4α-PDD)
Piperine (black pepper, long pepper)Polygodial (Dorrigo pepper)Probenecid Protons
RhTx
Rutamarin (Ruta graveolens )
Resiniferatoxin (RTX) (Euphorbia resinifera/pooissonii )Shogaols (ginger , Sichuan and melegueta peppers)
Tetrahydrocannabivarin (cannabis)
Thymol (thyme, oregano)Tinyatoxin (Euphorbia resinifera/pooissonii )
Tramadol Vanillin (vanilla)Zucapsaicin Blockers
See also: Receptor/signaling modulators • Ion channel modulators