Enoximone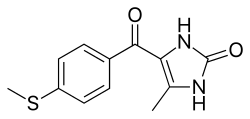 |
|
| Trade names | Perfan |
|---|
| AHFS/Drugs.com | International Drug Names |
|---|
Routes of
administration | Intravenous |
|---|
| ATC code | |
|---|
|
| Legal status |
- UK: POM (Prescription only)
|
|---|
|
| Bioavailability | 50% (oral) |
|---|
| Protein binding | 85% |
|---|
| Metabolism | Liver (oxidation) |
|---|
| Elimination half-life | 4 to 10 hours |
|---|
| Excretion | Renal (60 to 70%) |
|---|
|
4-Methyl-5-{[4-(methylsulfanyl)phenyl]carbonyl}-2,3-dihydro-1H-imidazol-2-one
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
| ECHA InfoCard | 100.419.380 |
|---|
|
| Formula | C12H12N2O2S |
|---|
| Molar mass | 248.30 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
| Melting point | 255 to 258 °C (491 to 496 °F) (decomposes) |
|---|
O=C(/C1=C(/NC(=O)N1)C)c2ccc(SC)cc2
|
InChI=1S/C12H12N2O2S/c1-7-10(14-12(16)13-7)11(15)8-3-5-9(17-2)6-4-8/h3-6H,1-2H3,(H2,13,14,16)  Y YKey:ZJKNESGOIKRXQY-UHFFFAOYSA-N  Y Y
|
| (verify) |
Enoximone (INN, trade name Perfan) is an imidazole phosphodiesterase inhibitor. It is used in the treatment of congestive heart failure and is selective for phosphodiesterase 3.[1]
References
- ^ Boldt J, Suttner S (September 2007). "Combined use of ultra-short acting beta-blocker esmolol and intravenous phosphodiesterase 3 inhibitor enoximone". Expert Opin Pharmacother. 8 (13): 2135–47. doi:10.1517/14656566.8.13.2135. PMID 17714066. S2CID 46021219.
External links
 Media related to Enoximone at Wikimedia Commons
Media related to Enoximone at Wikimedia Commons
Phosphodiesterase inhibitors |
|---|
| PDE1 | |
|---|
| PDE2 |
- BAY 60-7550
- Carbazeran
- EHNA
- Oxindole
- PDP
|
|---|
| PDE3 | |
|---|
| PDE4 |
- Apremilast
- Arofylline
- Atizoram
- Benafentrine
- Catramilast
- CC-1088
- CDP-840
- CGH-2466
- Cilomilast
- Cipamfylline
- Crisaborole
- Denbutylline
- Difamilast
- Drotaverine
- Ensifentrine
- Etazolate
- Filaminast
- Glaucine
- HT-0712
- ICI-63197
- Indimilast
- Irsogladine
- Lavamilast
- Lirimilast
- Lotamilast
- Luteolin
- Mesembrenone
- Mesembrine
- Mesopram
- Oglemilast
- Piclamilast
- Pumafentrine
- Revamilast
- Ro 20-1724
- Roflumilast
- Rolipram
- Ronomilast
- RPL-554
- RS-25344
- Tetomilast
- Tofimilast
- YM-976
- Zardaverine
|
|---|
| PDE5 |
- Acetildenafil
- Aildenafil
- Avanafil
- Beminafil
- Benzamidenafil
- Dasantafil
- Icariin
- Gisadenafil
- Homosildenafil
- Lodenafil
- Mirodenafil
- MY-5445
- Nitrosoprodenafil
- Norcarbodenafil
- SCH-51866
- Sildenafil
- Sulfoaildenafil
- T-0156
- Tadalafil
- Udenafil
- Vardenafil
|
|---|
| PDE7 | |
|---|
| PDE9 | |
|---|
| PDE10 |
- Balipodect
- Mardepodect
- MK-8189
- Papaverine
- TC-E 5005
- Tofisopam
|
|---|
| PDE11 | BC11-38 |
|---|
| Non-selective | |
|---|
| Unsorted | |
|---|
See also: Receptor/signaling modulators |
Cardiac stimulants excluding cardiac glycosides (C01C) |
|---|
Adrenergic and
dopaminergic agents | | Adrenergic agonists | |
|---|
| Dopamine agonists | |
|---|
| Both | |
|---|
| Unknown/ungrouped | |
|---|
|
|---|
| Phosphodiesterase inhibitors (PDE3I) | |
|---|
| Other cardiac stimulants |
- Angiotensinamide
- Levosimendan
- Omecamtiv mecarbil
- Pimobendan
- Xamoterol
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|

 Media related to Enoximone at Wikimedia Commons
Media related to Enoximone at Wikimedia Commons