Gold(I) chloride
-chlorid.png) | |
| Names | |
|---|---|
| IUPAC name
Gold(I) chloride
| |
| Other names
Aurous chloride
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.030.583 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
AuCl |
| Molar mass | 232.423 g/mol |
| Appearance | yellow solid[1] |
| Density | 7.6 g/cm3[2] |
| Melting point | 210 °C (410 °F; 483 K) (decomposition)[3] |
Solubility in water
|
Insoluble (cold water) Reacts (warm water)[2] |
| Solubility | Soluble in HCl, HBr organic solvents[2] |
Magnetic susceptibility (χ)
|
−67.0·10−6 cm3/mol[1] |
| Structure[4] | |
Crystal structure
|
Tetragonal, tI16 |
Space group
|
I41/amd, No. 141 |
Lattice constant
|
a = 6.734 Å, c = 8.674 Å
|
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
–34.7 kJ/mol[1] |
| Hazards | |
| GHS labelling: | |
Pictograms
|
 
|
Signal word
|
Danger |
Hazard statements
|
H314, H317 |
| NFPA 704 (fire diamond) | 
3
1
0 |
| Safety data sheet (SDS) | MSDS (Fisher) |
| Related compounds | |
Other anions
|
Gold(I) fluoride Gold(I) bromide Gold(I) iodide |
Other cations
|
Copper(I) chloride Silver chloride |
Related compounds
|
Gold(I,III) chloride Gold(III) chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Gold(I) chloride is a compound of gold and chlorine with the chemical formula AuCl. It is a metastable yellow solid that hydrolyses in warm water and decomposes to gold and chlorine gas at elevated temperatures. It has limited uses in organic chemistry as a Lewis acid.
History
Gold(I) chloride was first reported by Louis Jacques Thénard in 1814, who had carefully heated gold(III) chloride and isolated a yellow solid, claiming that it was the proto-muriate of gold. Joseph Proust disputed his claim, along with Thénard's other erroneous claims, such as of gold(I) oxide, later in the year.[5][6] However, gold(I) chloride was found to be a distinct compound from gold(III) chloride, as the synthesis was repeated by others, such as Hans Peter Jørgen Julius Thomsen. The structure was elucidated in 1974 by X-ray diffraction.[4][7]
Preparation
A modern synthesis entails the decomposition of gold(III) chloride under a chlorine atmosphere at 247 °C:[4]
- AuCl3 → AuCl + Cl2
Gold(III) chloride can also be heated in air at 185 °C to produce gold(I) chloride with careful temperature control.[7]
Structure
The structure of gold(I) chloride, which crystallizes in the tetragonal crystal system, consists of zig-zag chains of gold and chlorine, similar to gold(I) bromide and gold(I) iodide. The Au-Cl bond length is 2.36 Å, and the chain bends at the Cl with a bond angle of 92°.[4][8][9]
Reactions
Gold(I) chloride decomposes to gold metal and chlorine gas at around 210 °C:[3][10]
- 2AuCl → 2Au + Cl2
Although a region of stability exists at higher temperatures and appropriate chlorine vapour pressures (254 to 282 °C at PCl2 = 1 atm), the compound remains metastable at ambient conditions. In warm water, the compound dispropotionates to metallic gold and gold(III) chloride in an autoredox reaction:[2][4][11][12]
- 3 AuCl → 2 Au + AuCl3
At still higher temperatures, around 500 °C, all gold chlorides convert to gold even under a chlorine atmosphere. This conversion is key to the Miller process, which is widely used for the purification of gold.[10][13]
In hydrochloric acid, gold(I) chloride dissolves to form the colorless dichloroaurate(I) (AuCl2–) ion. At higher pH, this complex hydrolyses to gold(I)-hydroxo complexes and disproportionates to gold and gold(III).[14] However, the salts of the dichloroaurate(I) ion, such as the tetrabutylammonium salt, are prepared from the respective tetrachloroaurate(III) ion instead from gold(I) chloride.[15]
Reaction with potassium bromide yields potassium auric bromide and potassium chloride with separation of metallic gold:[2]
- 3 AuCl + 4 KBr → KAuBr4 + 2 Au + 3 KCl
Carbon monoxide reduces gold(I) chloride at 110 °C to form phosgene:
- 2AuCl + CO → 2Au + COCl2
However, under benzene, it forms carbonylchlorogold(I) (COAuCl) instead.[16][17]
Organogold(I) complexes
Various gold(I) chloride complexes are known, such as Ph3PAuCl and Me2SAuCl. However, the synthesis of these does not utilize gold(I) chloride, but instead goes through a direct reduction from Au(III) in the presence of the ligand. These complexes are the major gateways to other organogold(I) complexes.[18][19][20]
Applications
Gold(I) chloride mostly only has applications in organogold catalyzed organic reactions. It is used as a Lewis acid to activate alkenes, alkynes, and allenes. For example, acetylenic acids cycloisomerize in the presence of gold(I) chloride:[21][22]
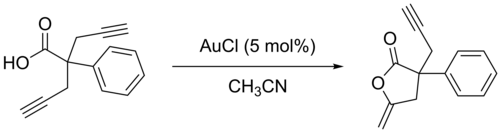
Safety
Generally, gold(I) compounds are less toxic than gold(III) compounds. However, gold(I) chloride is a skin sensitizer that may irritate the skin and eyes.[13][23]
References
- ^ a b c Haynes, W. M., ed. (2015). CRC Handbook of Chemistry and Physics (95th ed.). Boca Raton, Florida: CRC Press/Taylor & Francis. ISBN 978-1482208672.
- ^ a b c d e Pradyot Patnaik. Handbook of Inorganic Chemicals. McGraw-Hill, 2002, ISBN 0-07-049439-8
- ^ a b Ya-jie Zheng; Wei Guo; Meng Bai; Xing-wen Yang (2006). "Preparation of chloroauric acid and its thermal decomposition". The Chinese Journal of Nonferrous Metals (in Chinese). 16 (11): 1976–1982. Archived from the original on March 27, 2024.
- ^ a b c d e E. M. W. Janssen; J. C. W. Folmer; G. A. Wiegers (1974). "The preparation and crystal structure of gold monochloride, AuCl". Journal of the Less Common Metals. 38 (1): 71–76. doi:10.1016/0022-5088(74)90204-5.
- ^ Louis Jacques Thénard (1814). "Muriate d'Or". Traité de chimie élémentaire, théorique et pratique, Vol. 2 [Treatise on Elementary Chemistry, Theoretical and Practical, Volume 2] (in French). Lyon Public Library: Chez Crochard. pp. 594–597.
- ^ Joseph Proust (1814). "Seconde Lettre sur L'Incertitude de Quelques Oxidations". Journal de physique, de chimie, d'histoire naturelle et des arts (in French). 74: 321–342. Retrieved 13 February 2026.
- ^ a b Julius Thomsen (1876). "Darstellung und Eigenschaften der Chlor- und Bromverbindungen und des Oxyds des Goldes". Journal für Praktische Chemie (in German). 13 (1): 337–347. doi:10.1002/prac.18760130119.
- ^ Janssen, E.M.W.; Wiegers, G.A. (1978). "Crystal growth and the crystal structures of two modifications of gold monobromide, I-AuBr and P-AuBr". Journal of the Less Common Metals. 57 (2): P47–P57. doi:10.1016/0022-5088(78)90248-5. Retrieved 2026-02-13.
- ^ Tang, Zhongjia; Litvinchuk, A. P.; Lee, Hye-G.; Guloy, Arnold M. (1998-09-01). "Crystal Structure and Vibrational Spectra of a New Viologen Gold(I) Iodide". Inorganic Chemistry. 37 (19): 4752–4753. doi:10.1021/ic980141q. ISSN 0020-1669. Retrieved 2026-02-13.
- ^ a b Yiqin Chen; Xuezeng Tian; Wei Zeng; Xupeng Zhu; Hailong Hu; Huigao Duan (2015). "Vapor-phase preparation of gold nanocrystals by chloroauric acid pyrolysis". Journal of Colloid and Interface Science. 439. Elsevier: 21–27. Bibcode:2015JCIS..439...21C. doi:10.1016/j.jcis.2014.10.017. PMID 25463171.
- ^ Housecroft, C. E.; Sharpe, A. G. (2018). Inorganic Chemistry (5th ed.). Prentice Hall. p. 863. ISBN 978-1292134147.
- ^ E.M.W. Janssen; F. Pohlmann; G. A. Wiegers (1976). "The phase diagram of the gold-chlorine system". Journal of the Less Common Metals. 45 (2): 261–273. doi:10.1016/0022-5088(76)90272-1.
- ^ a b Renner, Hermann; Schlamp, Günther; Hollmann, Dieter; Lüschow, Hans Martin; Tews, Peter; Rothaut, Josef; Dermann, Klaus; Knödler, Alfons; Hecht, Christian; Schlott, Martin; Drieselmann, Ralf; Peter, Catrin; Schiele, Rainer (2000). "Gold, Gold Alloys, and Gold Compounds". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a12_499. ISBN 3527306730.
- ^ Christopher H. Gammons; Yunmei Yu; A. E. Williams-Jones (1997). "The disproportionation of gold(I) chloride complexes at 25 to 200°C". Geochimica et Cosmochimica Acta. 61 (10): 1971–1983. doi:10.1016/S0016-7037(97)00060-4.
- ^ Edwards, D. A. (1991). "Synthesis of Complex Halides Derived from Monohalides of Group‐IB". Inorganic Reactions and Methods. Vol. 4. Wiley. p. 127–133. doi:10.1002/9780470145180.ch72. ISBN 978-0-471-18657-1. Retrieved 2026-02-13.
- ^ T.A. Ryan; E.A. Seddon; K.R. Seddon; C. Ryan (1996). Phosgene And Related Carbonyl Halides. Elsevier Science. pp. 242–243. ISBN 9780080538808.
- ^ M. S. Kharasch; H. S. Isbell (1930). "The Chemistry of Organic Gold Compounds. I. Aurous Chloride Carbonyl and a Method of Linking Carbon to Carbon". Journal of the American Chemical Society. 52 (7): 2919–2927. doi:10.1021/ja01370a052.
- ^ Pierre Braunstein; Hans Lehner; Dominique Matt (1990). A Platinum-Gold Cluster: Chloro-1κCl-Bis(Triethylphosphine-1κP)Bis(Triphenylphosphine)-2κP, 3κP-Triangulo- Digold-Platinum(1 +) Trifluoromethanesulfonate. Inorganic Syntheses. Vol. 27. pp. 218–221. doi:10.1002/9780470132586.ch42.
- ^ Marie-Claude Brandys, Michael C. Jennings and Richard J. Puddephatt (2000). "Luminescent gold(I) macrocycles with diphosphine and 4,4-bipyridyl ligands". J. Chem. Soc., Dalton Trans. (24): 4601–4606. doi:10.1039/b005251p.
- ^ de Orbe, M. Elena; Echavarren, Antonio M. (2016). "Intermolecular [2+2] Cycloaddition of Alkynes with Alkenes Catalyzed by Gold(I)". Org. Synth. 93: 115. doi:10.15227/orgsyn.093.0115.
- ^ Li, Changkun; Wang, Jianbo (2009). "Gold Chloride". Encyclopedia of Reagents for Organic Synthesis. Chichester, UK: John Wiley & Sons, Ltd. doi:10.1002/047084289x.rn01001. ISBN 978-0-471-93623-7.
- ^ Genin, Emilie; Toullec, Patrick Yves; Antoniotti, Sylvain; Brancour, Célia; Genêt, Jean-Pierre; Michelet, Véronique (2006-03-01). "Room Temperature Au(I)-Catalyzed exo -Selective Cycloisomerization of Acetylenic Acids: An Entry to Functionalized γ-Lactones". Journal of the American Chemical Society. 128 (10): 3112–3113. doi:10.1021/ja056857j. ISSN 0002-7863.
- ^ "SAFETY DATA SHEET Gold(I) chloride". Fisher Scientific. ThermoFisher Scientific. Retrieved 13 February 2026.