Tantalum(V) chloride
 Tantalum, Ta; Chlorine, Cl
| |
| Names | |
|---|---|
| IUPAC names
Tantalum(V) chloride
Tantalum pentachloride | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.028.869 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
TaCl5 |
| Molar mass | 358.213 g/mol |
| Appearance | white monoclinic crystals[1] |
| Density | 3.68 g/cm3 |
| Melting point | 216 °C (421 °F; 489 K) |
| Boiling point | 239.4 °C (462.9 °F; 512.5 K) (decomposes) |
Solubility in water
|
reacts |
| Solubility | chloroform, CCl4 |
Magnetic susceptibility (χ)
|
+140.0×10−6 cm3/mol |
| Structure | |
Crystal structure
|
Monoclinic, mS72 |
Space group
|
C2/m, No. 12 |
| Thermochemistry | |
Std molar
entropy (S⦵298) |
221.75 J K−1 mol−1 |
Std enthalpy of
formation (ΔfH⦵298) |
−858.98 kJ/mol |
| Hazards | |
| GHS labelling: | |
Pictograms
|
 
|
Signal word
|
Danger |
Hazard statements
|
H302, H314, H335 |
Precautionary statements
|
P260, P264+P265, P270, P271, P280, P301+P317, P301+P330+P331, P302+P361+P354, P304+P340, P305+P351+P338, P305+P354+P338, P310, P316, P330, P363, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 
3
0
0 |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
1900 mg/kg (oral, rat) |
| Safety data sheet (SDS) | External SDS |
| Related compounds | |
Other anions
|
|
Other cations
|
|
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Tantalum(V) chloride, also known as tantalum pentachloride, is an inorganic compound with the formula TaCl5. It takes the form of a white powder, which is highly sensitive to moisture. It is synthesised and manipulated under anhydrous conditions, using air-free techniques.[2][3]
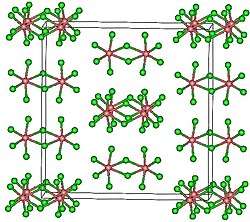
Structure
TaCl5 crystallizes in the monoclinic space group C2/m.[4] The ten chlorine atoms define a pair of octahedra that share a common edge. The tantalum atoms occupy the centres of the octahedra and are joined by two chlorine bridging ligands. The dimeric structure Ta2Cl10 is retained in non-complexing solvents and to a large extent in the molten state. In the vapour state, however, TaCl5 is monomeric. This monomer adopts a trigonal bipyramidal structure, like that of PCl5.[5]
Synthesis
Tantalum pentachloride can be prepared by treating powdered metallic tantalum with chlorine gas at between 170 and 250 °C. This reaction can also be performed using HCl at 400 °C.[6]
- 2 Ta + 5 Cl2 → 2 TaCl5
- 2 Ta + 10 HCl → 2 TaCl5 + 5 H2
It can also be prepared by a reaction between tantalum pentoxide and thionyl chloride at 240 °C.[3]
- Ta2O5 + 5 SOCl2 → 2 TaCl5 + 5 SO2
Tantalum pentachloride is commercially available, however samples can be contaminated with tantalum(V) oxytrichloride (TaOCl3), formed by hydrolysis. Samples contaminated with niobium pentachloride appear yellow.[3]
Reactions
TaCl5 is electrophilic. It forms adducts with a variety of Lewis bases. It serves as a Lewis acid in Friedel-Crafts reactions.[7]
Simple adducts
TaCl5 forms stable complexes with ethers:.[7][8]
TaCl5 also reacts with phosphorus pentachloride and phosphoryl chloride, the former as a chloride donor and the latter serves as a ligand, binding through the oxygen:[9]
- TaCl5 + PCl5 → [PCl4]+[TaCl6]−
- TaCl5 + OPCl3 → [TaCl5(OPCl3)]
Tantalum pentachloride reacts with tertiary amines to give adducts:[7]
- TaCl5 + 2 R3N → [TaCl5(NR3)]
Chloride displacement reactions
Tantalum pentachloride reacts at room temperature with an excess of triphenylphosphine oxide to give oxychlorides:
- TaCl5 + 3 OPPh3 → [TaOCl3(OPPh3)]x ...
Hydrolysis, alcoholysis, ammonolysis and related reactions
Anhydrous TaCl5 (Ta2Cl10) rapidly (but not violently) hydrolyses in water at room temperature.[10]
Tantalum pentachloride reacts with sodium ethoxide to give the pentaethoxide, which also exists as a dimer:[11]
- 5 NaOC2H5 + TaCl5 → Ta(OC2H5)5 + NaCl
A variety of related alkoxides and mixed chloro-alkoxides are known, e.g. Ta(OCH3)5 and Ta(OCH2CH3)3Cl2.[7]
Ammonium chloride gives the nitrido complex [TaCl5N]3−.[12]
Reduction
Reduction of tantalum(V) chloride gives anionic and neutral clusters including [Ta6Cl18]4− and [Ta6Cl14](H2O)4.[13]
References
- ^ Lide, David R., ed. (2006). CRC Handbook of Chemistry and Physics (87th ed.). Boca Raton, Florida: CRC Press. ISBN 0-8493-0487-3.
- ^ Clark, R. J. H.; Brown, D.; Bailar, J. C.; Emeléus, H. J.; Nyholm, Ronald (2013). The Chemistry of Vanadium, Niobium and Tantalum: Pergamon Texts in Inorganic Chemistry. Texts in Inorganic Chemistry (1. Aufl ed.). s.l.: Elsevier Reference Monographs. ISBN 978-1-4831-8170-7.
- ^ a b c G. Brauer (1963). "Niobium (V) and Tantalum (V) Chlorides". In G. Brauer (ed.). Handbook of Preparative Inorganic Chemistry, 2nd Ed. Vol. 2. NY, NY: Academic Press. pp. 1302–1305.
- ^ Rabe, Susanne; Müller, Ulrich (2000). "Crystal structure of tantalum pentachloride, (TaCl5)2". Z. Kristallogr. - New Cryst. Struct. 215 (1): 1–2. Bibcode:2000ZK....215....1R. doi:10.1515/ncrs-2000-0102.
- ^ Fairbrother, F. (1967). The Chemistry of Niobium and Tantalum. Elsevier.
- ^ Young, Ralph C.; Brubaker, Carl H. (1952). "Reaction of Tantalum with Hydrogen Chloride, Hydrogen Bromide and Tantalum Pentachloride; Action of Hydrogen on Tantalum Pentachloride". Journal of the American Chemical Society. 74 (19): 4967. Bibcode:1952JAChS..74.4967Y. doi:10.1021/ja01139a524.
- ^ a b c d Hubert-Pfalzgraf, Liliane G. (2005). "Niobium & Tantalum: Inorganic & Coordination Chemistry". Encyclopedia of Inorganic Chemistry. doi:10.1002/0470862106.ia153. ISBN 978-0-470-86078-6.
- ^ Cowley, A.; Fairbrother, F.; Scott, N. (1958). "The Halides of Niobium (Columbium) and Tantalum. Part V. Diethyl Ether Complexes of the Pentachlorides and Pentabromides; the Solubility of Tantalum Pentaiodide in Ether". J. Chem. Soc.: 3133–3137. doi:10.1039/JR9580003133.
- ^ Bullock, J. I.; Parrett, F. W.; Taylor, N. J. (1973). "Some Metal Halide–Phosphorus Halide–Alkyl Halide Complexes. Part II. Reactions with Niobium and Tantalum Pentachlorides and Tungsten Hexachloride". J. Chem. Soc., Dalton Trans. (5): 522–524. doi:10.1039/DT9730000522.
- ^ "Tantalum(V) chloride | Tantalum pentachloride | TaCl5". Ereztech. Retrieved 2025-12-03.
- ^ Schubert, U. (2003). "Sol–Gel Processing of Metal Compounds". In McCleverty, J. A.; Meyer, T. J. (eds.). Comprehensive Coordination Chemistry II. Reference Module in Chemistry, Molecular Sciences and Chemical Engineering. Vol. 7. Pergamon. pp. 629–656. doi:10.1016/B0-08-043748-6/06213-7. ISBN 978-0-12-409547-2.
- ^ Berg, Rolf W. (1992). "Progress in Niobium and Tantalum Coordination Chemistry". Coordination Chemistry Reviews. 113: 1–130. doi:10.1016/0010-8545(92)80074-2.
- ^ Duraisamy, Thirumalai; Hay, Daniel N. T.; Messerle, Louis (2014). "Octahedral Hexatantalum Halide Clusters". Inorganic Syntheses. Vol. 36. pp. 1–8. doi:10.1002/9781118744994.ch1. ISBN 9781118744994.
Further reading
- Ozin, G. A.; Walton, R. A. (1970). "Vibrational Spectra and Structures of the 1:1 Complexes of Niobium and Tantalum, Pentachlorides and Tantalum Pentabromide with Aceto-, Perdeuterioaceto-, and Propionitriles in the Solid and Solution States and a Vibrational Analysis of the Species MX5, NC·CY3 (Y = H or D)". J. Chem. Soc. A: 2236–2239. doi:10.1039/j19700002236.
- Đorđević, C.; Katović, V. (1970). "Co-ordination Complexes of Niobium and Tantalum. Part VIII. Complexes of Niobium(IV), Niobium(V), and Tantalum(V) with Mixed Oxo, Halogeno, Alkoxy, and 2,2′-Bipyridyl Ligands". J. Chem. Soc. A: 3382–3386. doi:10.1039/j19700003382.