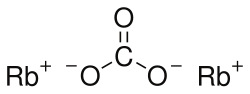
Rubidium carbonate
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Rubidium carbonate
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.008.666 |
PubChem CID
|
|
| RTECS number |
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
Rb2CO3 |
| Molar mass | 230.945 g/mol |
| Appearance | White powder, very hygroscopic |
| Melting point | 837 °C (1,539 °F; 1,110 K)[2] |
| Boiling point | 900 °C (1,650 °F; 1,170 K) (decomposes) |
Solubility in water
|
4500 g/L @20°C[1] |
Magnetic susceptibility (χ)
|
−75.4·10−6 cm3/mol |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Irritant |
| GHS labelling:[3] | |
Pictograms
|

|
Signal word
|
Warning |
Hazard statements
|
H315, H319, H335 |
Precautionary statements
|
P261, P264, P264+P265, P271, P280, P302+P352, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P403+P233, P405, P501 |
| Flash point | Non-flammable |
| Related compounds | |
Other cations
|
Lithium carbonate Sodium carbonate Potassium carbonate Caesium carbonate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Rubidium carbonate is an inorganic compound with the chemical formula Rb2CO3. It is a white, hygroscopic solid that readily dissolves in water.
Preparation
Rubidium carbonate can be prepared by reacting ammonium carbonate and rubidium hydroxide.[4]
Uses
It is used in glassmaking to enhance stability and durability whilst reducing conductivity.[5]
It is also used as a part of a catalyst to prepare short-chain alcohols from feed gas.
References
- ^ "Rubidium carbonate, 99%, Thermo Scientific Chemicals". Fisher Scientific. 1970-01-01. Retrieved 2025-05-27.
- ^ Patnaik, Pradyot (2002). Handbook of Inorganic Chemicals. McGraw-Hill. ISBN 0-07-049439-8.
- ^ PubChem. "Carbonic acid, rubidium salt (1:2)". pubchem.ncbi.nlm.nih.gov. Retrieved 2026-01-19.
- ^ Chisholm, Hugh, ed. (1911). . Encyclopædia Britannica. Vol. 23 (11th ed.). Cambridge University Press. p. 809.
- ^ Zhang, Zhifei; Li, Weilun; Zhang, Wenjuan; Wang, Chenyang; Zhao, Zhongwei; Choi, Yeonuk; Zhang, Han; Zhao, Tianyu; Sadri, Farzaneh (2026-02-15). "A review of rubidium: Resources, technologies, and applications". Desalination. 620 119612: 7. doi:10.1016/j.desal.2025.119612. ISSN 0011-9164.