Alizapride AHFS/Drugs.com International Drug Names Routes of Oral, IM, IV ATC code Legal status
In general: ℞ (Prescription only)
Elimination half-life 3 hours Excretion Renal
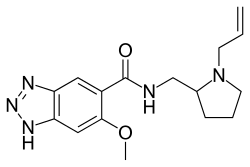
N -[(1-Allylpyrrolidin-2-yl)methyl]-6-methoxy-1H -benzo[d ][1,2,3]triazole-5-carboxamide
CAS Number PubChem CID DrugBank ChemSpider UNII KEGG ChEMBL CompTox Dashboard (EPA ) ECHA InfoCard 100.056.082 Formula C 16 H 21 N 5 O 2 Molar mass −1 3D model (JSmol)
C=CCN1CCCC1CNC(=O)c3cc2nn[nH]c2cc3OC
InChI=1S/C16H21N5O2/c1-3-6-21-7-4-5-11(21)10-17-16(22)12-8-13-14(19-20-18-13)9-15(12)23-2/h3,8-9,11H,1,4-7,10H2,2H3,(H,17,22)(H,18,19,20)
Y Key:KSEYRUGYKHXGFW-UHFFFAOYSA-N
Y (verify)
Alizapride (Litican , Plitican , Superan , Vergentan ) is a dopamine antagonist with prokinetic and antiemetic effects used in the treatment of nausea and vomiting, including postoperative nausea and vomiting. It is structurally related to metoclopramide and other benzamides .[ 1]
Mechanism
Alizapride blocks D2 dopamine receptors in the vomiting center.[ 2]
Since alizapride is able to cross the blood-brain barrier, adverse effects may include temporary extrapyramidal motor disorders such as acute dystonia and dyskinesia.[ 3]
It has a plasma half-life of 3 hours.[ 3]
Synthesis
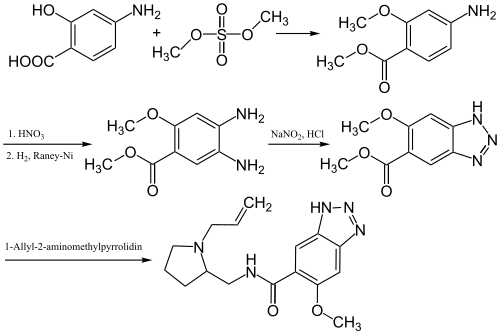
The synthesis of Alizapride happens in multiple steps:[ 4]
Synthesis of Alacepril 4-Aminosalicylic acid is first methylated using dimethyl sulfate . A nitro group is then introduced that is reduced using Raney nickel to afford an amino group. The two amino groups are then closed to a triazole ring using sodium nitrite and hydrochloric acid . This is then condensed with 1-allyl-2-aminomethylpyrrolidine to afford Alizapride.
References
^ Ballatori E, Roila F (September 2003). "Impact of nausea and vomiting on quality of life in cancer patients during chemotherapy" . Health and Quality of Life Outcomes . 1 : 46. doi:10.1186/1477-7525-1-46 212194 14521717 . ^ Online GL (October 17, 2016). "Anwendung, Wirkung, Nebenwirkungen" . Gelbe Liste Online (in German). Retrieved April 30, 2025 . ^ a b Geisslinger G, Menzel S, Gundermann T, Roth P (2020). Mutschler Arzneimittelwirkungen (11 ed.). Stuttgart: Wissenschaftliche Verlagsgesellschaft. p. 580. ISBN 978-3-8047-3663-4 . ^ Kleemann A, Engel J, Kutscher B, Reichert D (2014). Pharmaceutical Substances, 5th Edition: Syntheses, Patents and Applications of the most relevant APIs . Georg Thieme Verlag. p. 41. ISBN 978-3-13-179525-0 .
Antiemetics (A04)
5-HT3 serotonin ion 5-HT serotonin G-protein CB1 agonists D2 /D3 antagonists H1 antagonists mACh antagonists NK1 antagonists
Aprepitant Fosaprepitant
Maropitant
Netupitant
Rolapitant
Tradipitant Others
Drugs for functional gastrointestinal disorders (A03)
Drugs for
Antimuscarinics
Tertiary Quaternary
Phosphodiesterase Acting on
5-HT3 antagonists
5-HT4 agonists
Other
Alverine Caroverine
Chlorbenzoxamine
Diisopromine
Dimethylaminopropionylphenothiazine
Fenoverine
Fenpiprane
Idanpramine
Isometheptene
Mentha piperita
Phloroglucinol Pinaverium bromide
Proxazole
Simeticone Tiropramide
Trepibutone
Trimethyldiphenylpropylamine
Belladonna Propulsives
Primarily dopamine antagonists
5-HT4 agonists
Acotiamide
Ilebopride
Mosapride
Dopamine receptor modulators
D1 -like
Agonists
Benzazepines : 6-Br-APBFenoldopam SKF-38,393
SKF-77,434
SKF-81,297
SKF-82,958
SKF-83,959
Trepipam
Zelandopam Dihydrexidine derivatives : A-77636A-86929
Adrogolide (ABT-431, DAS-431)
Dihydrexidine
Dinapsoline
Dinoxyline
Doxanthrine Phenethylamines : BCO-001Deoxyepinephrine (N -methyldopamine, epinine)
Dipropyldopamine (DPDA)
Dopamine
Dopexamine
Etilevodopa
Ibopamine
L -DOPA (levodopa)Lu AE04621
Melevodopa
L -PhenylalanineL -TyrosineXP21279 Others : A-68930Apomorphine Isocorypalmine
Lu AF28996
Nuciferine
PF-06412562
PF-6649751
PF 6669571
Propylnorapomorphine
Rotigotine
SKF-89,145
SKF-89,626
Stepholidine
Tavapadon
Tetrahydropalmatine PAMs
Tetrahydroisoquinolines : DETQDPTQ
Glovadalen
Mevidalen Antagonists
Others : BerupipamEcopipam
EEDQ Metitepine (methiothepin)
Odapipam
Perlapine
SCH-23390
D2 -like
Agonists
Aminotetralins : 5-OH-DPAT7-OH-DPAT
8-OH-PBZI
CHF-1024
Nolomirole
Rotigotine
UH-232 Dihydrexidine derivatives : 2-OH-NPACiladopa
Dihydrexidine
Dinoxyline
N,N-Propyldihydrexidine Phenethylamines : Deoxyepinephrine (N-methyldopamine, epinine)Dipropyldopamine (DPDA)
Dopamine
Dopexamine
Etilevodopa
Ibopamine
L -DOPA (levodopa)Lu AE04621
L -PhenylalanineRU-24213
L -TyrosineMelevodopa
XP21279 Atypical antipsychotics : Alentemol (U-66444B)Aripiprazole (+sertraline)Aripiprazole lauroxil
Bifeprunox
Brexpiprazole
Brilaroxazine
Cariprazine
F-15063
Lumateperone
Norclozapine Others : 3-PPPA-412997
ABT-670
ABT-724
Adrafinil Aplindore
Apomorphine Arketamine
Armodafinil
BP-897
Captodiame
CP-226,269
Debenzergoline
Dizocilpine
Esketamine
Etrabamine
Flibanserin
7-Hydroxyropinirole (SK&F-89124)
Ketamine Lu AF28996
Matsupexole
Modafinil Naxagolide
OSU-6162
Pardoprunox
PD-128,907
PD-168,077
PF-219,061
PF-592,379
Phencyclidine Piribedil
Pramipexole
Preclamol
Propylnorapomorphine
Pukateine
Quinagolide
Quinelorane
Quinpirole
RDS-127
Ro10-5824
Ropinirole Roxindole
S32504
Salvinorin A
SKF-39315
SKF-83,959
Sumanirole
Talipexole
Umespirone
WAY-100,635
XC-130 Antagonists
Others : 3-PPPAlpiropride
Azapride
Bromerguride
Bromocriptine Buspirone Carmoxirole
Desmethoxyfallypride
EEDQ F-15063
Fallypride
Fananserin
Fenfluramine Iodobenzamide
Isocorypalmine
L-741,626
L-745,870
Levofenfluramine
LEK-8829
Metitepine (methiothepin)
N-Methylspiperone
Nafadotride
Nuciferine
Ordopidine
PNU-99,194
Pridopidine
Raclopride
Sarizotan
SB-277,011-A
Seridopidine
Sonepiprazole
Spiroxatrine
Stepholidine
SV-293
Tetrahydropalmatine Tiapride UH-232
XC-130
Yohimbine
See also: Receptor/signaling modulatorsAdrenergics Serotonergics Monoamine reuptake inhibitors Monoamine releasing agents Monoamine metabolism modulators Monoamine neurotoxins