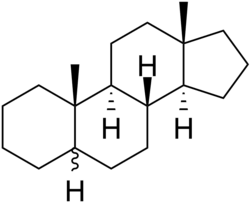
Androstane
|
| Names
|
| IUPAC name
5ξ-Androstane
|
Systematic IUPAC name
(3aS,3bS,5aΞ,9aS,9bS,11aS)-9a,11a-Dimethylhexadecahydro-1H-cyclopenta[a]phenanthrene |
| Other names
Etioallocholane; 10β,13β-Dimethylgonane
|
| Identifiers
|
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI
|
|
| ChemSpider
|
|
|
|
|
| UNII
|
|
|
|
|
InChI=1S/C19H32/c1-18-11-5-7-16(18)15-9-8-14-6-3-4-12-19(14,2)17(15)10-13-18/h14-17H,3-13H2,1-2H3/t14?,15-,16-,17-,18-,19-/m0/s1  Y YKey: QZLYKIGBANMMBK-FZFXZXLVSA-N  Y Y
|
C[C@@]12[C@](CCC2)([H])[C@]3([H])CCC4([H])CCCC[C@]4(C)[C@@]3([H])CC1 C3C1CCCC[C@@]1([C@H]2CC[C@]4([C@H]([C@@H]2C3)CCC4)C)C
|
| Properties
|
Chemical formula
|
C19H32
|
| Molar mass
|
260.465 g·mol−1
|
| Density
|
0.95 g/ml
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
|
Androstane is a C19 steroidal hydrocarbon with a gonane core. Androstane can exist as either of two isomers, known as 5α-androstane and 5β-androstane.
-
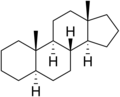
5α-Androstane
-
5β-Androstane
Pharmacology
5α-Androstane is reported to be effective as an androgen, in spite of having no oxygen containing functional groups.[1][2]
Androstanes
Androstanes are steroid derivatives with carbons present at positions 1 through 19.
See also
- Estrane and pregnane
- C19H32
References
Steroid classification |
|---|
| C17 | |
|---|
| C18 | |
|---|
| C19 | |
|---|
| C20 | |
|---|
| C21 |
- Pregnane: Dehydrogenated: Pregnene
- Pregnadiene
- Pregnatriene; Substituted: Pregnanediol
- Pregnanetriol
- Pregnenediol
- Pregnanedione
- Pregnenedione
- 5α-Pregnane
- 5β-Pregnane
|
|---|
| C23 | |
|---|
| C24 | |
|---|
| C27 | |
|---|
| Functional group |
- 17-Ketosteroid
- Hydroxysteroid
- Halogenated steroid
|
|---|
| Elements removed | |
|---|
| Elements replaced | |
|---|
Androgen receptor modulators |
|---|
| ARTooltip Androgen receptor | | Agonists | |
|---|
| SARMsTooltip Selective androgen receptor modulator |
- Nonsteroidal: 198RL26
- ACP-105
- AC-262,536
- Acetothiolutamide
- Acetoxolutamide
- Andarine (acetamidoxolutamide, androxolutamide, GTx-007, S-4)
- BMS-564,929
- DTIB
- Enobosarm (ostarine, MK-2866, GTx-024, S-22)
- FTBU-1
- GLPG-0492
- GSK2881078
- GSK-4336A
- GSK-8698
- LG121071 (LGD-121071)
- LGD-2226
- LGD-2941 (LGD-122941)
- LGD-3303
- LGD-4033
- LY305
- JNJ-26146900
- JNJ-28330835
- JNJ-37654032
- OPK-88004 (LY-2452473, TT-701)
- ORM-11984
- PF-06260414
- R-1
- RU-59063
- S-1
- S-23
- S-40503
- S-101479
- Vosilasarm
- Steroidal: EM-9017
- MK-0773
- S42
- TFM-4AS-1
- YK-11
|
|---|
| Antagonists |
- Steroidal: 7α-Thioprogesterone
- 7α-Thiospironolactone
- 7α-Thiomethylspironolactone
- 11α-Hydroxyprogesterone
- 15β-Hydroxycyproterone acetate
- Abiraterone
- Abiraterone acetate
- Allyltestosterone
- Benorterone
- BOMT
- Canrenoic acid
- Canrenone
- Chlormadinone acetate
- Clascoterone
- Clometerone
- Cyproheptadine
- Cyproterone
- Cyproterone acetate
- Delanterone
- Delmadinone acetate
- Dicirenone
- Dienogest
- Drospirenone
- DU-41165
- Edogestrone
- EM-4350
- EM-5854
- EM-5855
- EM-6537
- Epitestosterone
- Galeterone
- Guggulsterone
- Ludaterone
- Medrogestone
- Megestrol acetate
- Mespirenone
- Metogest
- Mexrenone
- Mifepristone
- Nomegestrol acetate
- Nordinone
- Osaterone
- Osaterone acetate
- Oxendolone
- Potassium canrenoate
- Promegestone
- Prorenone
- Rosterolone
- RU-15328
- SC-5233 (spirolactone)
- Spironolactone
- Spirorenone
- Spiroxasone
- Topterone
- Trimegestone
- Trimethyltrienolone (R-2956)
- Zanoterone
|
|---|
|
|---|
| GPRC6A | |
|---|
- See also
- Receptor/signaling modulators
- Androgens and antiandrogens
- Estrogen receptor modulators
- Progesterone receptor modulators
- List of androgens and anabolic steroids
|

 5α-Androstane
5α-Androstane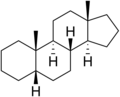 5β-Androstane
5β-Androstane