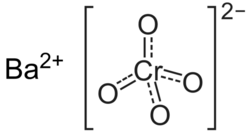
Barium chromate
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Barium chromate
| |
| Other names
Barium chromate oxide,
Chromic acid, (BaCrO4), barium salt (1:1), barium tetraoxochromate(VI) | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.030.587 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1564 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
BaCrO4 |
| Molar mass | 253.37 g/mol |
| Appearance | yellow powder, darkens upon heating |
| Density | 4.498 g/cm3 |
| Melting point | 210 °C (410 °F; 483 K) (decomposes) |
Solubility in water
|
0.2775 mg/100 mL (20 °C) |
Solubility product (Ksp)
|
1.17 × 10−10[1] |
| Solubility | soluble in strong acids |
| Structure | |
Crystal structure
|
orthorhombic |
| Hazards | |
| GHS labelling: | |
Pictograms
|
    
|
Signal word
|
Danger |
Hazard statements
|
H272, H301, H302, H317, H332, H350, H410 |
Precautionary statements
|
P201, P202, P210, P220, P221, P261, P264, P270, P271, P272, P273, P280, P281, P301+P310, P301+P312, P302+P352, P304+P312, P304+P340, P308+P313, P312, P321, P330, P333+P313, P363, P370+P378, P391, P405, P501 |
| NFPA 704 (fire diamond) | 
3
0
3 |
| Safety data sheet (SDS) | External MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Barium chromate is an inorganic compound with the chemical formula BaCrO4. It is a yellow solid, an oxidizing agent that produces a green flame when heated, a result of the barium ions.
Properties
It is very insoluble in water, but soluble in acids:
- 2 BaCrO4 + 2 H+ → 2 Ba2+ + Cr2O72− + H2O Ksp = [Ba2+][CrO42−] = 2.1 × 10−10
Preparation
Barium chromate can be precipitated by reacting a soluble barium salt such as barium hydroxide with a chromate such as potassium chromate:
- Ba(OH)2 + K2CrO4 → BaCrO4↓ + 2 KOH
Single-crystalline ABO4 type nanorods can be prepared by template synthesis. Nanoparticles are allowed to grow in the pores of alumina membranes of various sizes. The varying sizes of the pores allow the growth to be controlled and cause the shapes to be reproducible. The alumina is then dissolved, leaving the nanoparticles behind intact. The synthesis can be carried out at room temperature.[2]
Uses
Barium chromate has been found to be useful in many capacities. The compound is often used as a carrier for the chromium ions.
Electroplating
Barium chromate acts as a sulfate scavenger in chromium electroplating baths.[3] Over time, the chromium concentration of the bath decreases until it is no longer functional. Adding barium chromate improves the lifetime of the bath by increasing the chromic acid concentration.
Barium chromate is used as a corrosion inhibitive pigment on zinc-alloy electroplating surfaces.[4]
Oxidizing agent
Barium chromate is an oxidizing agent, making it useful as a burn rate modifier in pyrotechnic compositions. It is especially useful in delay compositions such as delay fuses.[5]
Pigment
Barium has been used to color paints. The pigment known as lemon yellow often contained barium chromate mixed with lead sulfate.[6] Due to its moderate tinting strength lemon yellow was not employed very frequently in oil painting.[7] Pierre-Auguste Renoir and Claude Monet are known to have painted with lemon yellow.[8]
Other
When mixed with solid fumaric acid, barium chromate can be used in the removal of impurities and residual moisture from dry-cleaning solvents or from petroleum fuels.[9]
Barium chromate is also used in the composition of a catalyst for alkane dehydrogenation.[10]
Reactions
Barium chromate reacts with barium hydroxide in the presence of sodium azide to form barium chromate(V). The reaction releases oxygen and water.
- 4 BaCrO4 + 2 Ba(OH)2 2 Ba3(CrO4)2 + O2↑ + 2 H2O↑
History
The first naturally occurring barium chromate mineral was found in the country of Jordan. The brown crystals found perched on host rocks were named hashemite in honor of the Hashemite Kingdom of Jordan. Hashemite crystals range in color from light yellowish-brown to a darker greenish-brown and are usually less than 1 mm in length.[11]
Hashemite crystals are not composed of pure barium chromate but instead contain some small sulfur content as well. The different crystals contain a range of sulfur impurities ranging from the more pure dark crystals, Ba1.00(Cr0.93, S0.07)1.00O4, to the less pure light crystals, Ba1.00(Cr0.64, S0.36)1.00O4.[12]
Hashemite was found to be an isostructural chromate analog of baryte, BaSO4.[13]
Safety
Barium chromate is toxic. Chromates, when pulverized and inhaled, are carcinogens.
In 2010, a study was conducted on four hexavalent chromium compounds to test the carcinogenic effects of chromium. The chromium ions accumulate in the bronchial bifurcation sites, settling into the tissue and inducing tumors. It was discovered that barium chromate is both genotoxic and cytotoxic. The cytotoxicity was determined to most likely be a result of the genotoxicity, but the cause of the genotoxicity is yet unknown.[14]
References
- ^ John Rumble (June 18, 2018). CRC Handbook of Chemistry and Physics (99th ed.). CRC Press. pp. 4–47. ISBN 978-1138561632.
- ^ Mao, Yuanbing; Wong, Stanislaus S. (2004) "General, Room-Temperature Method for the Synthesis and well as Arrays of Single-Crystalline ABO4− Type Nanorods." J. Am. Chem. Soc. 126(46), 15245-15252.
- ^ Jones, Allen R; Meng, John; Newboy, Kenneth R. (1993) Use of barium chromate as a sulfate scavenger in chromium electroplating baths" US Patent No. 5207890 A.
- ^ Fountoulakis, Stavros G; Humayan, Arif; Lezzi, Robert A. (1985) "Electroplated product and method" EP Patent No. 0140564 A2.
- ^ Espagnacq, Andre; Morand, Philippe. (1997) "Pyrotechnic composition for delay fuses" EP Patent No 0630876 B1.
- ^ Douma, M. curator. (2008) Pigments through the Ages. Institute for Dynamic Educational Development. http://www.webexhibits.org/pigments/
- ^ Kühn, H. and Curran, M., Strontium, Barium and Calcium Chromates, in Artists' Pigments. A Handbook of Their History and Characteristics, Vol. 1: Feller, R.L. (Ed.) Oxford University Press 1986, p. 205 – 207.
- ^ Lemon yellow, ColourLex
- ^ Jackson, Herman R. (1993) "SOlid fumaric acid-solid barium chromate catalyst for removing impurities and residual moisture and method for its use" US Patent No. 5154726 A.
- ^ Ruettinger, Wolfgang; Jacubinas, Richard. (2013) "Chromia Alumina Catalysts for Alkane Dehydrogenation" US Patent No. 20130072739 A1.
- ^ Hauff, Phoebe L; Foord, Eugene E; Rosenblum, Sam; and Hakki, Walid. (1983) "Hashemite, Ba(Cr,S)O4, a new mineral from Jordan." American Mineralogist, 69, 1223-1225.
- ^ Hauff, Phoebe L; Foord, Eugene E; Rosenblum, Sam; and Hakki, Walid. (1983) "Hashemite, Ba(Cr,S)O4, a new mineral from Jordan." American Mineralogist, 69, 1223-1225.
- ^ Hauff, Phoebe L; Foord, Eugene E; Rosenblum, Sam; and Hakki, Walid. (1983) "Hashemite, Ba(Cr,S)O4, a new mineral from Jordan." American Mineralogist, 69, 1223-1225.
- ^ Wise, Sandra S; Holmes, Amie L; Qin, Qin; Xie, Hong; Kafsifis, Spiros P; Thompson, W Douglas; Wise, John Pierce Sr. (2010) "Comparative Genotoxicity and Cytotoxicity of Four Haxevalent Chromium Compounds in Human Bronchial Cells." Chem. Res. Toxicol. 23, 365-372.
Further study
- Kühn, H. and Curran, M., Strontium, Barium and Calcium Chromates, in Artists' Pigments. A Handbook of Their History and Characteristics, Vol. 1: Feller, R.L. (Ed.) Oxford University Press 1986, p. 205 – 207.