Barium sulfite
 | |
| Names | |
|---|---|
| IUPAC name
Barium sulfite
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.029.193 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
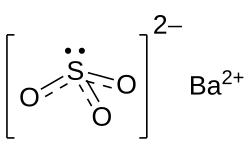
Chemical formula
|
BaSO3 |
| Molar mass | 217.391 g/mol |
| Appearance | white monoclinic crystals |
| Density | 4.44 g/cm3 |
| Melting point | decomposes |
Solubility in water
|
0.0011 g/100 mL |
| Solubility | insoluble in ethanol[1] |
| Related compounds | |
Other anions
|
Barium sulfate Barium fluoride Barium chloride Barium bromide Barium iodide |
Other cations
|
Calcium sulfite Magnesium sulfite |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Barium sulfite is the inorganic compound with the chemical formula BaSO3. It is a white powder that finds few applications.
Occurrence
It is an intermediate in the carbothermal reduction of barium sulfate to barium sulfide:[2]
- BaSO4 + CO → BaSO3 + CO2
It also occurs as an intermediate phase in the aqueous oxidation of BaS to BaSO4 by weathering in slag.[3]
Uses
Barium sulfite is used in the hydrolysis of dialdehyde starch by sulfurous acid yielding glyoxal and erythrose.[4] The barium sulfite complexes with the glyoxal, allowing for higher reagent concentrations without loss of yield.[4]
References
- ^ Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, Florida: CRC Press, pp. 4–45, ISBN 0-8493-0594-2
- ^ Kresse, Robert; Baudis, Ulrich; Jäger, Paul; Riechers, H. Hermann; Wagner, Heinz; Winkler, Jochen; Wolf, Hans Uwe (2007). "Barium and Barium Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a03_325.pub2. ISBN 978-3527306732.
- ^ Braithwaite, R. S. W.; Kampf, A. R.; Pritchard, R. G.; Lamb, R. P. H. (1993-06-01). "The occurrence of thiosulfates and other unstable sulfur species as natural weathering products of old smelting slags". Mineralogy and Petrology. 47 (2): 255–261. doi:10.1007/BF01161570. ISSN 1438-1168.
- ^ a b Wilham, C. A.; McGuire, T. A.; Cleve, J. W. Van; Otey, F. H.; Mehltretter, C. L. (2002-05-01). "Hydrolysis of Dialdehyde Starch. Glyoxal and Erythrose Production in Sulfurous Acid-Barium Sulfite Solutions". ACS Publications. doi:10.1021/i360001a014. Retrieved 2026-01-24.