Barium bromate
 | |
| Identifiers | |
|---|---|
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.034.304 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 2719 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
Ba(BrO3)2 or BaBr2O6 |
| Molar mass | 393.13 |
| Appearance | White crystalline powder |
| Hazards | |
| GHS labelling: | |
Pictograms
|
 
|
Signal word
|
Warning |
Hazard statements
|
H272, H302, H332 |
Precautionary statements
|
P210, P220, P221, P261, P264, P270, P271, P280, P301+P312, P304+P312, P304+P340, P312, P330, P370+P378, P501 |
| Related compounds | |
Other cations
|
calcium bromate strontium bromate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
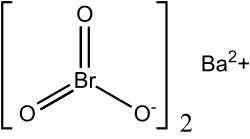
Barium bromate is a chemical compound composed of the barium ion and the bromate ion, with the chemical formula of Ba(BrO3)2.
Preparation
Barium bromate can be prepared by reacting potassium bromate or sodium bromate with barium chloride:[1]
- 2 KBrO3 + BaCl2 → Ba(BrO3)2 + 2 KCl