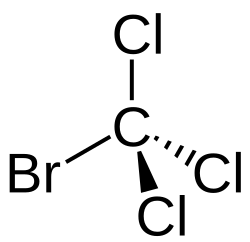
Bromotrichloromethane
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Bromo(trichloro)methane | |
| Other names
Carbon bromotrichloride
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.000.806 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
CBrCl3 |
| Molar mass | 198.27 g·mol−1 |
| Appearance | colorless liquid |
| Density | 2.401 g/cm³ |
| Melting point | 21 °C (70 °F; 294 K) |
| Boiling point | 105 °C (221 °F; 378 K) |
Solubility in water
|
practically insoluble |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|

|
Signal word
|
Warning |
| Flash point | 32.6 °C |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Bromotrichloromethane is a tetrahalomethane with the chemical formula CBrCl3. This is a halomethane containing one bromine atom and three chlorine atoms.[2][3] Bromotrichloromethane is a volatile, non-flammable, colorless liquid with a pungent odor that is practically insoluble in water but miscible with organic solvents.[4]
Synthesis
Bromotrichloromethane can be obtained by treating carbon tetrachloride with hydrogen bromide or aluminum bromide.[5] It can also be obtained by reacting trichloromethane with bromine at 225 to 400 °C or potassium trichloroacetate with bromine at 120 °C.[6]
- Cl3CCO2K + Br2 → CBrCl3 + CO2 + KBr
Physical properties
Uses
Bromotrichloromethane is used as a chain transfer agent for the radical polymerization of methacrylates and as a brominating agent.[7]
It is used for the addition of Br-CCl3 across double bonds.[2]
References
- ^ "Bromotrichloromethane". Sigma Aldrich. Retrieved 5 September 2025.
- ^ a b Shinokubo, Hiroshi; Oshima, Koichiro; Gu, Zhenhua; Zakarian, Armen; Castiglione, Davide; Pace, Vittorio (2025). "Bromotrichloromethane (CBRCL 3 )". Encyclopedia of Reagents for Organic Synthesis. pp. 1–10. doi:10.1002/047084289X.rn00250.pub3. ISBN 978-0-471-93623-7.
- ^ Crich, David (30 May 2013). Reagents for Radical and Radical Ion Chemistry. John Wiley & Sons. p. 12. ISBN 978-1-118-63489-9. Retrieved 5 September 2025.
- ^ Anderson, Bruce; Peyster, Ann de; Gad, Shayne C.; Hakkinen, P. J. Bert; Kamrin, Michael; Locey, Betty; Mehendale, Harihara M.; Pope, Carey; Shugart, Lee (31 May 2005). Encyclopedia of Toxicology. Elsevier. ISBN 978-0-08-054800-5. Retrieved 5 September 2025.
- ^ Skell, P. S; Baxter, H. N; Tanko, J. M (1 January 1986). "Reactions of BrCl with alkyl radicals". Tetrahedron Letters. 27 (43): 5181–5184. doi:10.1016/S0040-4039(00)85163-1. ISSN 0040-4039. Retrieved 5 September 2025.
- ^ Gilchrist, Thomas L. (1995). Comprehensive Organic Functional Group Transformations (1 ed.). Oxford: Elsevier. p. 221. ISBN 0-08-042704-9. Retrieved 5 September 2025.
- ^ "Bromtrichlormethan". gestis.dguv.de. Retrieved 5 September 2025.