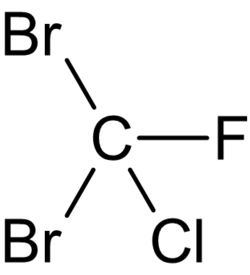
Dibromochlorofluoromethane
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Dibromo(chloro)fluoromethane | |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.209.965 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
CBr2ClF |
| Molar mass | 226.27 g·mol−1 |
| Density | 2.6 g/cm3 |
| Boiling point | 78.9 °C (174.0 °F; 352.0 K) |
| Hazards | |
| Flash point | 1.5±18.4 °C |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Dibromochlorofluoromethane is a tetrahalomethane with the chemical formula CBr2ClF.[1][2] This is an organic compound containing two bromine atoms, one fluorine atom, and one chlorine atom attached to the methane backbone.[3][4]
Synthesis
It can be prepared by reacting silver chlorobromoacetate with bromine at 180-260 °C, or by brominating fluorodichloromethane at 650 °C.[5]
- CBrClFCO2Ag + Br2 → CBr2ClF + CO2 + AgBr
Uses
It can be used to introduce chlorofluoromethyl groups into organic compounds.[6]
References
- ^ Lide, David R. (9 March 1995). CRC Handbook of Chemistry and Physics: A Ready-reference Book of Chemical and Physical Data. CRC Press. p. 3-157. ISBN 978-0-8493-0595-5. Retrieved 8 September 2025.
- ^ "NCATS Inxight Drugs — DIBROMOCHLOROFLUOROMETHANE". drugs.ncats.io. Retrieved 8 September 2025.
- ^ "Dibromochlorofluoromethane". NIST. Retrieved 8 September 2025.
- ^ Luo, Yu-Ran (9 March 2007). Comprehensive Handbook of Chemical Bond Energies. CRC Press. p. 234. ISBN 978-1-4200-0728-2. Retrieved 8 September 2025.
- ^ Katritzky, Alan R.; Gilchrist, Thomas L.; Meth-Cohn, Otto; Rees, Charles Wayne (21 March 2003). Comprehensive Organic Functional Group Transformations. Elsevier. p. 225. ISBN 978-0-08-042704-1. Retrieved 8 September 2025.
- ^ Kvíčala, Jaroslav; Štambaský, Jan; Skalický, Martin; Paleta, Oldřich (1 October 2005). "Preparation of fluorohalomethylmagnesium halides using highly active magnesium metal and their reactions". Journal of Fluorine Chemistry. 126 (9): 1390–1395. Bibcode:2005JFluC.126.1390K. doi:10.1016/j.jfluchem.2005.07.016. ISSN 0022-1139. Retrieved 8 September 2025.