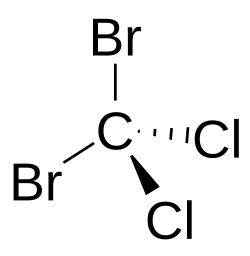
Dibromodichloromethane
 | |
| Names | |
|---|---|
| Other names
Carbon dibromide dichloride
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.008.938 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
CBr2Cl2 |
| Molar mass | 242.72 g·mol−1 |
| Appearance | solid |
| Density | 2.433 g/cm³ |
| Melting point | 38 °C (100 °F; 311 K) |
| Boiling point | 130.2 °C (266.4 °F; 403.3 K) |
Solubility in water
|
insoluble |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|

|
Signal word
|
Warning |
| Flash point | 32.6 °C |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Dibromodichloromethane is a tetrahalomethane with the chemical formula CBr2Cl2.[2] This is a halomethane containing two bromine atoms and two chlorine atoms attached to the methane backbone.[3][4]
Synthesis
Dibromodichloromethane can be obtained by treating tetrachloromethane with bromine trifluoride in the presence of aluminum chloride or boron tribromide.
Also it can be obtained, by the reaction of carbon tetrachloride and aluminum tribromide.[5]
Physical properties
Dibromodichloromethane forms a solid,[6] insoluble in water. It reacts with alkenes by addition of bromine and dichlorobromomethyl.[7]
Dibromodichloromethane is a potentially toxic compound.[8]
References
- ^ "594-18-3・Dibromodichloromethane・329-47401・327-47402[Detail Information]". Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation. Retrieved 3 September 2025.
- ^ "Dibromodichloromethane". NIST. Retrieved 3 September 2025.
- ^ Katritzky, Alan R.; Gilchrist, Thomas L.; Meth-Cohn, Otto; Rees, Charles Wayne (21 March 2003). Comprehensive Organic Functional Group Transformations. Elsevier. p. 226. ISBN 978-0-08-042704-1. Retrieved 3 September 2025.
- ^ Toxicological Evaluation of Chemical Interactions. National Institutes of Health, National Institute of Environmental Health Sciences. 1994. p. 141. Retrieved 3 September 2025.
- ^ Tanaka, Ryuichi; Zhèng, Shì-Qín; Kawaguchi, Kenji; Tanaka, Takehide (1 January 1980). "Nucleophilic attack on halogeno(phenyl)acetylenes by halide ions". Journal of the Chemical Society, Perkin Transactions 2 (11): 1714–1720. doi:10.1039/P29800001714. ISSN 1364-5471. Retrieved 3 September 2025.
- ^ Williams, Donald E.; Smyth, Charles P. (1 May 1962). "The Dielectric Relaxation of Dibromodichloromethane, Succinonitrile and Several Camphor Derivatives in the Solid State". Journal of the American Chemical Society. 84 (10): 1808–1812. doi:10.1021/ja00869a007. ISSN 0002-7863. Retrieved 3 September 2025.
- ^ Schmoldt, A.; Benthe, H. F.; Haberland, G. (1 September 1975). "Digitoxin metabolism by rat liver microsomes". Biochemical Pharmacology. 24 (17): 1639–1641. doi:10.1002/anie.201704074.
- ^ "Human Metabolome Database: Showing metabocard for Dibromodichloromethane (HMDB0061733)". HMDB. Retrieved 3 September 2025.