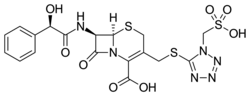
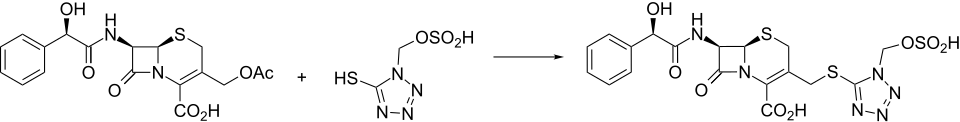Cefonicid AHFS/Drugs.com Micromedex Detailed Consumer Information MedlinePlus a601206 ATC code
(6R ,7R )-7-[(2R )-2-hydroxy-2-phenylacetyl)amino]-8-oxo-
CAS Number PubChem CID ChemSpider UNII KEGG ChEBI ChEMBL CompTox Dashboard (EPA ) Formula C 18 H 18 N 6 O 8 S 3 Molar mass −1 3D model (JSmol)
O=C2N1/C(=C(\CS[C@@H]1[C@@H]2NC(=O)[C@H](O)c3ccccc3)CSc4nnnn4CS(=O)(=O)O)C(=O)O
InChI=1S/C18H18N6O8S3/c25-13(9-4-2-1-3-5-9)14(26)19-11-15(27)24-12(17(28)29)10(6-33-16(11)24)7-34-18-20-21-22-23(18)8-35(30,31)32/h1-5,11,13,16,25H,6-8H2,(H,19,26)(H,28,29)(H,30,31,32)/t11-,13-,16-/m1/s1
Y Key:DYAIAHUQIPBDIP-AXAPSJFSSA-N
Y (verify)
Cefonicide (or cefonicid ) is a cephalosporin antibiotic.[ 1]
It has a density of 1.92g/cm3 .
Synthesis
Injectable semi-synthetic cephalosporin antibiotic related to cefamandole , q.v.
Cefonicid synthesis:[ 2] [ 3] [ 4] Cefonicid is synthesized conveniently by nucleophilic displacement of the 3-acetoxy moiety of 1 with the appropriately substituted tetrazole thiole 2 . The mandelic acid amide C-7 side chain is reminiscent of cefamandole .
See also
References
^ Saltiel E, Brogden RN (September 1986). "Cefonicid. A review of its antibacterial activity, pharmacological properties and therapeutic use". Drugs . 32 (3): 222– 59. doi:10.2165/00003495-198632030-00002 . PMID 3530703 . S2CID 243051006 . ^ DE 2611270 , Berges, David Alan, "Cephalosporin-Derivate [Cephalosporin derivatives]", published 1976-09-30, assigned to SmithKline Corp. ^ D. A. Berges, U.S. patent 4,048,311
^ U.S. patent 4,093,723 U.S. patent 4,159,373
Antibacterials active on the cell wall and envelope (J01C-J01D)
β-lactams(inhibit synthesisD -alanyl-D -alanine
Penicillins (Penams)
Narrow
β-lactamase sensitive β-lactamase resistant
Extended
Aminopenicillins (3rd generation) Carboxypenicillins (4th generation) Ureidopenicillins (4th generation) Other
Carbapenems / Penems
Carbapenems (Ertapenem
Antipseudomonal (Doripenem
Imipenem Meropenem )Biapenem‡
Panipenem)
Penems (Faropenem
Ritipenem§
Sulopenem) Cephems
1st generation 2nd generation 3rd generation 4th generation
Cefepime Cefozopran‡
Cefpirome
Cefquinome‡ 5th generation
Ceftaroline fosamil
Ceftolozane
Ceftobiprole Siderophore Veterinary
Ceftiofur
Cefquinome
Cefovecin
Monobactams
Aztreonam Tigemonam‡
Carumonam‡
Nocardicin A‡ β-lactamase inhibitors
Penam (Sulbactam
Tazobactam Enmetazobactam)
Clavam (Clavulanic acid )
non-β-lactam (Avibactam
Durlobactam
Relebactam
Taniborbactam
Vaborbactam) Combinations
Amoxicillin/clavulanic acid#
Ampicillin/flucloxacillin
Ampicillin/sulbactam (Sultamicillin)
Aztreonam/avibactam
Benzathine benzylpenicillin/procaine benzylpenicillin
Cefepime/enmetazobactam
Cefepime/sulbactam
Cefoperazone/sulbactam
Ceftazidime/avibactam
Ceftolozane/tazobactam
Imipenem/cilastatin#
Imipenem/cilastatin/relebactam
Meropenem/vaborbactam
Panipenem/betamipron
Piperacillin/tazobactam
Sulbactam/durlobactam
Sulopenem/probenecid
Ticarcillin/clavulanic acid
Polypeptides
Lipopeptides
Insert into bacterial cell wall causing perforation and depolarization: Daptomycin
Surfactin Other
Inhibits PG elongation and crosslinking: Ramoplanin§
Intracellular
Inhibit PG subunit synthesis and transport: NAM synthesis inhibition
DADAL/AR inhibitors
bactoprenol inhibitors
Other
Hydrolyze NAM-NAG
Tyrothricin
Isoniazid # Teixobactin
# WHO-EM‡ Withdrawn from marketClinical trials:
† Phase III§ Never to phase III