Tazobactam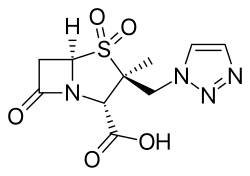 |
 |
|
| AHFS/Drugs.com | International Drug Names |
|---|
| License data |
|
|---|
Pregnancy
category | |
|---|
Routes of
administration | Intravenous |
|---|
| ATC code | |
|---|
|
| Legal status |
- In general: ℞ (Prescription only)
|
|---|
|
(2S,3S,5R)-3-Methyl-7-oxo-3-(1H-1,2,3-triazol-1-ylmethyl)-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid 4,4-dioxide
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEBI | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
| ECHA InfoCard | 100.108.321 |
|---|
|
| Formula | C10H12N4O5S |
|---|
| Molar mass | 300.29 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
O=S2(=O)[C@]([C@@H](N1C(=O)C[C@H]12)C(=O)O)(Cn3nncc3)C
|
InChI=1S/C10H12N4O5S/c1-10(5-13-3-2-11-12-13)8(9(16)17)14-6(15)4-7(14)20(10,18)19/h2-3,7-8H,4-5H2,1H3,(H,16,17)/t7-,8+,10+/m1/s1  Y YKey:LPQZKKCYTLCDGQ-WEDXCCLWSA-N  Y Y
|
 N N Y (what is this?) (verify) Y (what is this?) (verify) |
Tazobactam is a pharmaceutical drug that inhibits the action of bacterial β-lactamases, especially those belonging to the SHV-1 and TEM groups. It is commonly used as its sodium salt, tazobactam sodium.
Tazobactam is combined with the extended spectrum β-lactam antibiotic piperacillin in the drug piperacillin/tazobactam, used in infections due to Pseudomonas aeruginosa. Tazobactam broadens the spectrum of piperacillin by making it effective against organisms that express β-lactamase and would normally degrade piperacillin.[1]
Tazobactam was patented in 1982 and came into medical use in 1992.[2]
See also
References
Antibacterials active on the cell wall and envelope (J01C-J01D) |
|---|
β-lactams
(inhibit synthesis
of peptidoglycan
layer of bacterial
cell wall by binding
to and inhibiting
PBPs, a group of
D-alanyl-D-alanine
transpeptidases) | | Penicillins (Penams) | Narrow
spectrum | β-lactamase sensitive
(1st generation) | |
|---|
β-lactamase resistant
(2nd generation) | |
|---|
|
|---|
Extended
spectrum | | Aminopenicillins (3rd generation) | |
|---|
| Carboxypenicillins (4th generation) | |
|---|
| Ureidopenicillins (4th generation) | |
|---|
| Other | |
|---|
|
|---|
|
|---|
| Carbapenems / Penems |
- Carbapenems (Ertapenem
- Antipseudomonal (Doripenem
- Imipenem
- Meropenem)
- Biapenem‡
- Panipenem)
- Penems (Faropenem
- Ritipenem§
- Sulopenem)
|
|---|
Cephems
Cephalosporins
Cephamycins
Carbacephems | | 1st generation | |
|---|
| 2nd generation | |
|---|
| 3rd generation | |
|---|
| 4th generation |
- Cefepime
- Cefozopran‡
- Cefpirome
- Cefquinome‡
|
|---|
| 5th generation |
- Ceftaroline fosamil
- Ceftolozane
- Ceftobiprole
|
|---|
| Siderophore | |
|---|
| Veterinary |
- Ceftiofur
- Cefquinome
- Cefovecin
|
|---|
|
|---|
| Monobactams |
- Aztreonam
- Tigemonam‡
- Carumonam‡
- Nocardicin A‡
|
|---|
| β-lactamase inhibitors |
- Penam (Sulbactam
- Tazobactam
- Enmetazobactam)
- Clavam (Clavulanic acid)
- non-β-lactam (Avibactam
- Durlobactam
- Relebactam
- Taniborbactam
- Vaborbactam)
|
|---|
| Combinations |
- Amoxicillin/clavulanic acid#
- Ampicillin/flucloxacillin
- Ampicillin/sulbactam (Sultamicillin)
- Aztreonam/avibactam
- Benzathine benzylpenicillin/procaine benzylpenicillin
- Cefepime/enmetazobactam
- Cefepime/sulbactam
- Cefoperazone/sulbactam
- Ceftazidime/avibactam
- Ceftolozane/tazobactam
- Imipenem/cilastatin#
- Imipenem/cilastatin/relebactam
- Meropenem/vaborbactam
- Panipenem/betamipron
- Piperacillin/tazobactam
- Sulbactam/durlobactam
- Sulopenem/probenecid
- Ticarcillin/clavulanic acid
|
|---|
|
|---|
| Polypeptides | | Lipopeptides |
- Insert into bacterial cell wall causing perforation and depolarization: Daptomycin
- Surfactin
|
|---|
| Other |
- Inhibits PG elongation and crosslinking: Ramoplanin§
|
|---|
|
|---|
| Intracellular |
- Inhibit PG subunit synthesis and transport: NAM synthesis inhibition
- DADAL/AR inhibitors
- bactoprenol inhibitors
|
|---|
| Other |
- Hydrolyze NAM-NAG
- Tyrothricin
- Isoniazid#
- Teixobactin
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|

