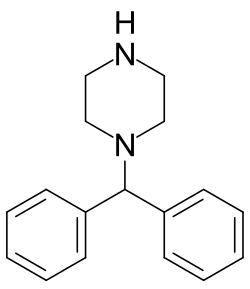
Diphenylmethylpiperazine
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1-(Diphenylmethyl)piperazine | |
| Other names
1-benzhydrylpiperazine; Norcyclizine
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.011.516 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C17H20N2 |
| Molar mass | 252.361 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Diphenylmethylpiperazine, also known as benzhydrylpiperazine, is a chemical compound and piperazine derivative. It features a piperazine ring with a benzhydryl (diphenylmethyl) group bound to one of the nitrogens.[1][2] Cyclizine, an antihistamine and anticholinergic drug used to treat motion sickness, is the methylated derivative of diphenylmethylpiperazine, and the antihistamine cinnarizine is another derivative. 1-Benzhydrylpiperazine has been described as having "amphetamine-like effects".[3]
References
- ^ Calderon SN (2011). Nonpeptidic delta (delta) opioid agonists and antagonists of the diarylmethylpiperazine class: what have we learned?. Topics in Current Chemistry. Vol. 299. pp. 121–140. Bibcode:2011chop.book..121C. doi:10.1007/128_2010_83. ISBN 978-3-642-18106-1. PMID 21630509.
{{cite book}}:|journal=ignored (help) - ^ Kaczor A, Matosiuk D (2002). "Non-peptide opioid receptor ligands - recent advances. Part I - agonists". Curr Med Chem. 9 (17): 1567–1589. doi:10.2174/0929867023369394. PMID 12171553.
- ^ Arslan, Zeynep; Okuroğlu, Eda; Şenol, Halil; Türkmen, Zeynep (2024). "1-Benzhydryl-piperazine: Isolation, structure determination, and in silico studies for a novel potential narcotic agent detected in sports supplements". Journal of Food Composition and Analysis. 135 106682. doi:10.1016/j.jfca.2024.106682.