Fusaric acid
Names
Preferred IUPAC name
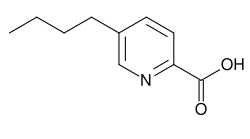
5-Butylpyridine-2-carboxylic acid
Other names
5-Butylpicolinic acid
Identifiers
CAS Number
3D model (JSmol)
ChemSpider
ECHA InfoCard
100.007.859
EC Number
KEGG
MeSH
D005669
UNII
InChI=1S/C10H13NO2/c1-2-3-4-8-5-6-9(10(12)13)11-7-8/h5-7H,2-4H2,1H3,(H,12,13)
N Key: DGMPVYSXXIOGJY-UHFFFAOYSA-N
N InChI=1/C10H13NO2/c1-2-3-4-8-5-6-9(10(12)13)11-7-8/h5-7H,2-4H2,1H3,(H,12,13)
Key: DGMPVYSXXIOGJY-UHFFFAOYAD
Properties
Chemical formula
C 10 H 13 N O 2
Molar mass
−1
Melting point
97 to 98 °C (207 to 208 °F; 370 to 371 K)
Related compounds
Related compounds
picolinic acid
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Fusaric acid is a picolinic acid derivative and an antibiotic (wilting agent) first isolated from the fungus Fusarium heterosporium .[ 1]
It is typically isolated from various Fusarium species, and has been proposed for a various therapeutic applications. However, it is primarily used as a research tool.
Its mechanism of action is not well understood. It likely inhibits dopamine beta-hydroxylase (the enzyme that converts dopamine to norepinephrine). It may also have other actions, such as the inhibition of cell proliferation and DNA synthesis. Fusaric acid and analogues also reported as quorum sensing inhibitors.[ 2]
It is used to make bupicomide.
References
External links
Monoamine metabolism modulators
Non-specific
AAAD Tooltip Aromatic L-amino acid decarboxylase MAO Tooltip Monoamine oxidase
Inhibitors: MAO-A-selective: AmiflamineBazinaprine
Befloxatone
Brofaromine
Cimoxatone
Clorgiline CX157 (Tyrima)
Eprobemide
Esuprone
Harmala alkaloids (e.g., harmine , harmaline , harman , norharman , tetrahydroharmine, 6-methoxyharman)
Methylene blue
Metralindole
Minaprine Moclobemide Pirlindole
Sercloremine
Tetrindole
Toloxatone
Inhibitors: MAO-B selective: AdarigilineAlmoxatone
D -DeprenylDesmethylselegiline
Ethanol 4-Fluorodeprenyl
4-Fluoroselegiline
Ladostigil
Lazabemide
Milacemide
Mofegiline
Nicotine Pargyline ‡ Rasagiline Safinamide
Selegiline (L -Deprenyl) Sembragiline
Tisolagiline
Vafidemstat
2-chloro-2-phenylethylamine
Phenethylamines(dopamine , epinephrine ,
PAH Tooltip Phenylalanine hydroxylase
Inhibitors: 3,4-Dihydroxystyreneα-Methylphenylalanine TH Tooltip Tyrosine hydroxylase DBH Tooltip Dopamine beta-hydroxylase
Substrates→Products: Dopamine →Norepinephrine (Noradrenaline) PNMT Tooltip Phenylethanolamine N-methyltransferase
Inhibitors: CGS-19281ASKF-64139
SKF-7698 COMT Tooltip Catechol-O-methyl transferase
Tryptamines(serotonin , melatonin )
TPH Tooltip Tryptophan hydroxylase AANAT Tooltip Serotonin N-acetyl transferase ASMT Tooltip Acetylserotonin O-methyltransferase
Histamine
HDC Tooltip Histidine decarboxylase
Inhibitors: Catechin Alpha-Fluoromethylhistidine
Histidine methyl ester
Meciadanol
Naringenin Tritoqualine HNMT Tooltip Histamine N-methyltransferase
Substrates→Products: Histamine →N-Methylhistamine DAO Tooltip Diamine oxidase
See also: Receptor/signaling modulators • Adrenergics • Dopaminergics • Melatonergics • Serotonergics • Monoamine reuptake inhibitors • Monoamine releasing agents • Monoamine neurotoxins
Authority control databases
 Media related to Fusaric acid at Wikimedia Commons
Media related to Fusaric acid at Wikimedia Commons