Minaprine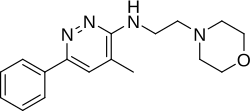 |
|
| AHFS/Drugs.com | International Drug Names |
|---|
Routes of
administration | Oral |
|---|
| ATC code | |
|---|
|
| Legal status |
- BR: Class C1 (Other controlled substances)[1]
- In general: ℞ (Prescription only)
|
|---|
|
| Elimination half-life | 2-2.5 hours |
|---|
|
4-methyl-N-(2-morpholin-4-ylethyl)-6-phenylpyridazin-3-amine
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
| ECHA InfoCard | 100.043.012 |
|---|
|
| Formula | C17H22N4O |
|---|
| Molar mass | 298.390 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
CC1=CC(=NN=C1NCCN2CCOCC2)C3=CC=CC=C3
|
InChI=1S/C17H22N4O/c1-14-13-16(15-5-3-2-4-6-15)19-20-17(14)18-7-8-21-9-11-22-12-10-21/h2-6,13H,7-12H2,1H3,(H,18,20)  Y YKey:LDMWSLGGVTVJPG-UHFFFAOYSA-N  Y Y
|
Minaprine (INN, USAN, BAN; brand names Brantur, Cantor) is a monoamine oxidase inhibitor antidepressant drug that was used in France for the treatment of depression until it was withdrawn from the market in 1996 because it caused convulsions.[2][3]
A study found that it acts as a reversible inhibitor of MAO-A (RIMA) in rats.[4] It has also been found to weakly inhibit acetylcholinesterase in rat brain (striatum) homogenates.[5]
It has demonstrated significant antibiotic activity against M. chelonae and M. abscessus in tests with antibiotic resistant bacteria.[6]
Synthesis
The first synthesis of minaprine was disclosed in patents published in 1979.[7]
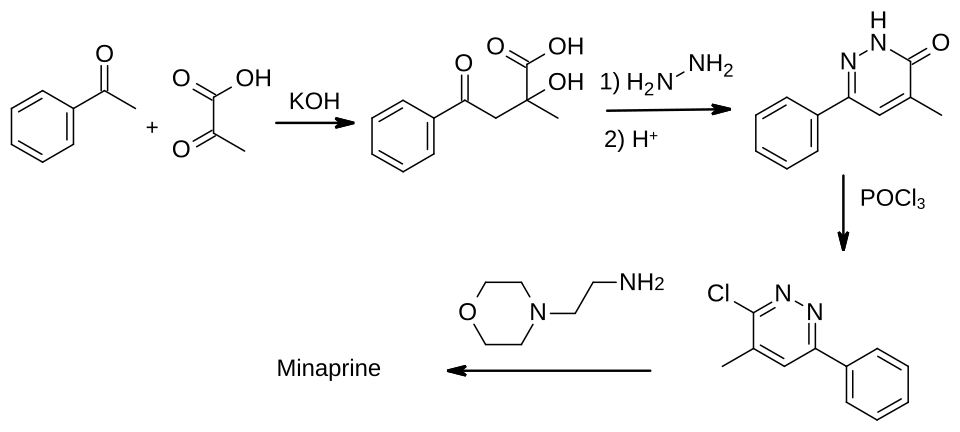
The final step is the reaction between a chloro-substituted pyridazine and the primary amine group of a morpholine derivative.[7][8] The required pyridazine can be made by the reaction of acetophenone and pyruvic acid, followed by ring formation using hydrazine, giving a pyrazidinone. Treatment of this with phosphoryl chloride converts it to the required chloro derivative.[2]
References
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ a b Wermuth CG, Schlewer G, Bourguignon JJ, Maghioros G, Bouchet MJ, Moire C, et al. (March 1989). "3-aminopyridazine derivatives with atypical antidepressant, serotonergic, and dopaminergic activities". Journal of Medicinal Chemistry. 32 (3): 528–537. doi:10.1021/jm00123a004. PMID 2563772.
- ^ Fung M, Thornton A, Mybeck K, Wu JH, Hornbuckle K, Muniz E (1 January 2001). "Evaluation of the Characteristics of Safety Withdrawal of Prescription Drugs from Worldwide Pharmaceutical Markets-1960 to 1999". Therapeutic Innovation & Regulatory Science. 35 (1): 293–317. doi:10.1177/009286150103500134. S2CID 73036562.
- ^ Kan JP, Mouget-Goniot C, Worms P, Biziere K (March 1986). "Effect of the antidepressant minaprine on both forms of monoamine oxidase in the rat". Biochemical Pharmacology. 35 (6): 973–978. doi:10.1016/0006-2952(86)90085-7. PMID 3954800.
- ^ Contreras JM, Rival YM, Chayer S, Bourguignon JJ, Wermuth CG (February 1999). "Aminopyridazines as acetylcholinesterase inhibitors". Journal of Medicinal Chemistry. 42 (4): 730–741. doi:10.1021/jm981101z. PMID 10052979.
- ^ Chopra S, Matsuyama K, Hutson C, Madrid P (July 2011). "Identification of antimicrobial activity among FDA-approved drugs for combating Mycobacterium abscessus and Mycobacterium chelonae". The Journal of Antimicrobial Chemotherapy. 66 (7): 1533–1536. doi:10.1093/jac/dkr154. PMID 21486854.
- ^ a b US patent 4169158, Henri Laborit, "Pyridazine derivatives in alleviating depressive states", issued 1979-09-25, assigned to CM Industries, SA
- ^ "Minaprine". Pharmaceutical Substances. Thieme. Archived from the original on 2022-12-12. Retrieved 2024-07-21.
Antidepressants (N06A) |
|---|
Specific reuptake inhibitors or receptor modulators |
|---|
| SSRIsTooltip Selective serotonin reuptake inhibitors | |
|---|
| SNRIsTooltip Serotonin–norepinephrine reuptake inhibitors | |
|---|
| NRIsTooltip Norepinephrine reuptake inhibitors | |
|---|
| NDRIsTooltip Norepinephrine–dopamine reuptake inhibitors | |
|---|
| NaSSAsTooltip Noradrenergic and specific serotonergic antidepressants | |
|---|
| SARIsTooltip Serotonin antagonist and reuptake inhibitors | |
|---|
| SMSTooltip Serotonin modulator and stimulators | |
|---|
| Others | |
|---|
|
|
Tricyclic and tetracyclic antidepressants |
|---|
| TCAsTooltip Tricyclic antidepressants | |
|---|
| TeCAsTooltip Tetracyclic antidepressants | |
|---|
| Others | |
|---|
|
|
Monoamine oxidase inhibitors |
|---|
| Non-selective | |
|---|
| MAOATooltip Monoamine oxidase A-selective | |
|---|
| MAOBTooltip Monoamine oxidase B-selective | |
|---|
|
|
|
|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
|
|---|
Enzyme
(modulators) | | ChATTooltip Choline acetyltransferase |
- Inhibitors: 1-(-Benzoylethyl)pyridinium
- 2-(α-Naphthoyl)ethyltrimethylammonium
- 3-Chloro-4-stillbazole
- 4-(1-Naphthylvinyl)pyridine
- Acetylseco hemicholinium-3
- Acryloylcholine
- AF64A
- B115
- BETA
- CM-54,903
- N,N-Dimethylaminoethylacrylate
- N,N-Dimethylaminoethylchloroacetate
|
|---|
| AChETooltip Acetylcholinesterase | |
|---|
| BChETooltip Butyrylcholinesterase | |
|---|
|
|---|
Transporter
(modulators) | | CHTTooltip Choline transporter |
- Inhibitors: Hemicholinium-3 (hemicholine)
- Triethylcholine
|
|---|
| VAChTTooltip Vesicular acetylcholine transporter | |
|---|
|
|---|
Release
(modulators) | | Inhibitors |
- Others: Bungarotoxins (β-bungarotoxin, γ-bungarotoxin)
|
|---|
| Enhancers |
- LPHNTooltip Latrophilin agonists: α-Latrotoxin
- Others: Atracotoxins (e.g., robustoxin, versutoxin)
- Crotoxin
|
|---|
|
|---|
- See also
- Receptor/signaling modulators
- Muscarinic acetylcholine receptor modulators
- Nicotinic acetylcholine receptor modulators
|
Monoamine metabolism modulators |
|---|
| Non-specific | | AAADTooltip Aromatic L-amino acid decarboxylase | |
|---|
| MAOTooltip Monoamine oxidase | |
- Inhibitors: MAO-A-selective: Amiflamine
- Bazinaprine
- Befloxatone
- Brofaromine
- Cimoxatone
- Clorgiline
- CX157 (Tyrima)
- Eprobemide
- Esuprone
- Harmala alkaloids (e.g., harmine, harmaline, harman, norharman, tetrahydroharmine, 6-methoxyharman)
- Methylene blue
- Metralindole
- Minaprine
- Moclobemide
- Pirlindole
- Sercloremine
- Tetrindole
- Toloxatone
|
- Inhibitors: MAO-B selective: Adarigiline
- Almoxatone
- D-Deprenyl
- Desmethylselegiline
- Ethanol
- 4-Fluorodeprenyl
- 4-Fluoroselegiline
- Ladostigil
- Lazabemide
- Milacemide
- Mofegiline
- Nicotine
- Pargyline‡
- Rasagiline
- Safinamide
- Selegiline (L-Deprenyl)
- Sembragiline
- Tisolagiline
- Vafidemstat
- 2-chloro-2-phenylethylamine
|
|
|---|
|
|---|
Phenethylamines
(dopamine, epinephrine,
norepinephrine) | | PAHTooltip Phenylalanine hydroxylase |
- Inhibitors: 3,4-Dihydroxystyrene
- α-Methylphenylalanine
|
|---|
| THTooltip Tyrosine hydroxylase | |
|---|
| DBHTooltip Dopamine beta-hydroxylase |
- Substrates→Products: Dopamine→Norepinephrine (Noradrenaline)
|
|---|
| PNMTTooltip Phenylethanolamine N-methyltransferase |
- Inhibitors: CGS-19281A
- SKF-64139
- SKF-7698
|
|---|
| COMTTooltip Catechol-O-methyl transferase | |
|---|
|
|---|
Tryptamines
(serotonin, melatonin) | | TPHTooltip Tryptophan hydroxylase | |
|---|
| AANATTooltip Serotonin N-acetyl transferase | |
|---|
| ASMTTooltip Acetylserotonin O-methyltransferase | |
|---|
|
|---|
| Histamine | | HDCTooltip Histidine decarboxylase |
- Inhibitors: Catechin
- Alpha-Fluoromethylhistidine
- Histidine methyl ester
- Meciadanol
- Naringenin
- Tritoqualine
|
|---|
| HNMTTooltip Histamine N-methyltransferase |
- Substrates→Products: Histamine→N-Methylhistamine
|
|---|
| DAOTooltip Diamine oxidase | |
|---|
|
|---|
See also: Receptor/signaling modulators • Adrenergics • Dopaminergics • Melatonergics • Serotonergics • Monoamine reuptake inhibitors • Monoamine releasing agents • Monoamine neurotoxins |

