Quinalizarin
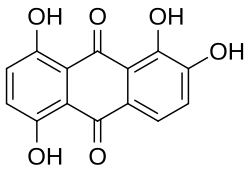
Skeletal formula of quinalizarin
Ball-and-stick model
Names
Preferred IUPAC name
1,2,5,8-Tetrahydroxyanthracene-9,10-dione
Identifiers
CAS Number
3D model (JSmol)
ChEMBL
ChemSpider
ECHA InfoCard
100.001.243
UNII
InChI=1S/C14H8O6/c15-6-3-4-7(16)11-10(6)12(18)5-1-2-8(17)13(19)9(5)14(11)20/h1-4,15-17,19H
N Key: VBHKTXLEJZIDJF-UHFFFAOYSA-N
N InChI=1/C14H8O6/c15-6-3-4-7(16)11-10(6)12(18)5-1-2-8(17)13(19)9(5)14(11)20/h1-4,15-17,19H
Key: VBHKTXLEJZIDJF-UHFFFAOYAM
C1=CC(=C(C2=C1C(=O)C3=C(C=CC(=C3C2=O)O)O)O)O
Properties
Chemical formula
C 14 H 8 O 6
Molar mass
−1
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Quinalizarin or 1,2,5,8-tetrahydroxyanthraquinone is an organic compound with formula C12 H4 (OH)4 (CO)2 . It is one of many tetrahydroxyanthraquinone isomers, formally derived from anthraquinone by replacement of four hydrogen atoms by hydroxyl (OH) groups at the 1, 2, 5, and 8 positions.
Quinalizarin is an inhibitor of the enzyme protein kinase CK2. It is more potent and selective than emodin .[ 1] [ 2] [ 3]
See also
References
^ Cozza, G.; Mazzorana, M.; Papinutto, E.; Bain, J.; Elliott, M.; di Maira, G.; Gianoncelli, A.; Pagano, M. A.; Sarno, S.; Ruzzene, M.; Battistutta, R.; Meggio, F.; Moro, S.; Zagotto, G.; Pinna, L. A. (2009). "Quinalizarin as a Potent, Selective and Cell-Permeable Inhibitor of Protein Kinase CK2" (PDF) . The Biochemical Journal . 421 (3): 387– 395. doi:10.1042/BJ20090069 . hdl:2262/45362 . PMID 19432557 . ^ Schneider J, Huh MM, Bradlow HL, Fishman J (April 1984). "Antiestrogen action of 2-hydroxyestrone on MCF-7 human breast cancer cells" . J. Biol. Chem . 259 (8): 4840– 5. doi:10.1016/S0021-9258(17)42922-X 6325410 . ^ Schütze N, Vollmer G, Knuppen R (April 1994). "Catecholestrogens are agonists of estrogen receptor dependent gene expression in MCF-7 cells". J. Steroid Biochem. Mol. Biol . 48 (5– 6): 453– 61. doi:10.1016/0960-0760(94)90193-7 . PMID 8180106 .
Monoamine metabolism modulators
Non-specific
AAAD Tooltip Aromatic L-amino acid decarboxylase MAO Tooltip Monoamine oxidase
Inhibitors: MAO-A-selective: AmiflamineBazinaprine
Befloxatone
Brofaromine
Cimoxatone
Clorgiline CX157 (Tyrima)
Eprobemide
Esuprone
Harmala alkaloids (e.g., harmine , harmaline , harman , norharman , tetrahydroharmine, 6-methoxyharman)
Methylene blue
Metralindole
Minaprine Moclobemide Pirlindole
Sercloremine
Tetrindole
Toloxatone
Inhibitors: MAO-B selective: AdarigilineAlmoxatone
D -DeprenylDesmethylselegiline
Ethanol 4-Fluorodeprenyl
4-Fluoroselegiline
Ladostigil
Lazabemide
Milacemide
Mofegiline
Nicotine Pargyline ‡ Rasagiline Safinamide
Selegiline (L -Deprenyl) Sembragiline
Tisolagiline
Vafidemstat
2-chloro-2-phenylethylamine
Phenethylamines(dopamine , epinephrine ,
PAH Tooltip Phenylalanine hydroxylase
Inhibitors: 3,4-Dihydroxystyreneα-Methylphenylalanine TH Tooltip Tyrosine hydroxylase DBH Tooltip Dopamine beta-hydroxylase
Substrates→Products: Dopamine →Norepinephrine (Noradrenaline) PNMT Tooltip Phenylethanolamine N-methyltransferase
Inhibitors: CGS-19281ASKF-64139
SKF-7698 COMT Tooltip Catechol-O-methyl transferase
Tryptamines(serotonin , melatonin )
TPH Tooltip Tryptophan hydroxylase AANAT Tooltip Serotonin N-acetyl transferase ASMT Tooltip Acetylserotonin O-methyltransferase
Histamine
HDC Tooltip Histidine decarboxylase
Inhibitors: Catechin Alpha-Fluoromethylhistidine
Histidine methyl ester
Meciadanol
Naringenin Tritoqualine HNMT Tooltip Histamine N-methyltransferase
Substrates→Products: Histamine →N-Methylhistamine DAO Tooltip Diamine oxidase
See also: Receptor/signaling modulators • Adrenergics • Dopaminergics • Melatonergics • Serotonergics • Monoamine reuptake inhibitors • Monoamine releasing agents • Monoamine neurotoxins