Pazufloxacin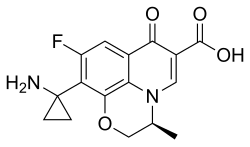 |
|
| AHFS/Drugs.com | International Drug Names |
|---|
| ATC code | |
|---|
|
(3R)-10-(1-aminocyclopropyl)-9-fluoro-3-methyl-7-oxo-1H,7H- [1,3]oxazino[5,4,3-ij]quinoline-carboxylic acid
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| CompTox Dashboard (EPA) | |
|---|
| ECHA InfoCard | 100.162.962 |
|---|
|
| Formula | C16H15FN2O4 |
|---|
| Molar mass | 318.304 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
C[C@H]1COC2=C3N1C=C(C(=O)C3=CC(=C2C4(CC4)N)F)C(=O)O
|
InChI=1S/C16H15FN2O4/c1-7-6-23-14-11(16(18)2-3-16)10(17)4-8-12(14)19(7)5-9(13(8)20)15(21)22/h4-5,7H,2-3,6,18H2,1H3,(H,21,22)/t7-/m0/s1 Key:XAGMUUZPGZWTRP-ZETCQYMHSA-N
|
| (verify) |
Pazufloxacin (INN) is a fluoroquinolone antibiotic.[1] It is sold in Japan under the brand names Pasil and Pazucross.
See also
References
- ^ Rubinstein E (2001). "History of quinolones and their side effects". Chemotherapy. 47 Suppl 3 (3): 3–8, discussion 44-8. doi:10.1159/000057838. PMID 11549783. S2CID 21890070.
Antibacterials that inhibit nucleic acid (J01E, J01M) |
|---|
Antifolates
(inhibit bacterial
purine metabolism,
thereby inhibiting
DNA and RNA
synthesis) | | DHFR inhibitor | |
|---|
Sulfonamides
(DHPS inhibitor) | | Short-acting | |
|---|
| Intermediate-acting | |
|---|
| Long-acting | |
|---|
| Other/ungrouped | |
|---|
|
|---|
| Combinations | |
|---|
| Other DHPS inhibitors | |
|---|
|
|---|
Quinolones
(inhibit bacterial
topoisomerase
and/or DNA gyrase,
thereby inhibiting
DNA replication) | | 1st generation | |
|---|
| Fluoroquinolones | | 2nd generation | |
|---|
| 3rd generation | |
|---|
| 4th generation | |
|---|
| Veterinary | |
|---|
|
|---|
| Newer non-fluorinated | |
|---|
| Related (DG) | |
|---|
|
|---|
Anaerobic DNA
inhibitors | | Nitroimidazole derivatives | |
|---|
|
|---|
| RNA synthesis | Rifamycins/
RNA polymerase | |
|---|
| Lipiarmycins | |
|---|
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
