Sulfametoxydiazine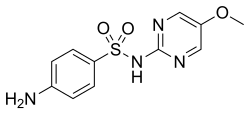 |
|
| ATC code | |
|---|
|
4-amino-N-(5-methoxy-2-pyrimidinyl)benzenesulfonamide
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEBI | |
|---|
| ChEMBL | |
|---|
| NIAID ChemDB | |
|---|
| CompTox Dashboard (EPA) | |
|---|
| ECHA InfoCard | 100.010.438 |
|---|
|
| Formula | C11H12N4O3S |
|---|
| Molar mass | 280.30 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
O=S(=O)(Nc1ncc(OC)cn1)c2ccc(N)cc2
|
InChI=1S/C11H12N4O3S/c1-18-9-6-13-11(14-7-9)15-19(16,17)10-4-2-8(12)3-5-10/h2-7H,12H2,1H3,(H,13,14,15)  Y YKey:GPTONYMQFTZPKC-UHFFFAOYSA-N  Y Y
|
 N N Y (what is this?) (verify) Y (what is this?) (verify) |
Sulfametoxydiazine (INN) or sulfamethoxydiazine (USAN: sulfameter) is a long-acting sulfonamide antibacterial.[1] It is used as a leprostatic agent and in the treatment of urinary tract infections.[2]
Sulfamethoxydiazine is also used to treat and prevent diseases in animals. Because of its relatively long persistence, sulfamethoxydiazine residue can be detected in meat, dairy, and eggs, and is considered hazardous to human health. The United States and Japan both prohibit sulfamethoxydiazine residue in food, whereas the Codex Alimentarius Commission states that the maximum limit for sulfonamides in animal tissues is 100 μg/kg.[1]
References
- ^ a b Wu Y, Yu S, Yu F, Yan N, Qu L, Zhang H (October 2011). "Chemiluminescence enzyme immunoassay for the determination of sulfamethoxydiazine". Spectrochimica Acta Part A. 81 (1): 544–547. Bibcode:2011AcSpA..81..544W. doi:10.1016/j.saa.2011.06.047. PMID 21795101.
- ^ Burros HM, Gillenwater JY (July 1965). "Clinical Experience with Sulfamethoxydiazine* in Urinary Tract Infections". The Journal of Urology. 94 (1): 86–88. doi:10.1016/S0022-5347(17)63576-6. PMID 14319481.
Antibacterials that inhibit nucleic acid (J01E, J01M) |
|---|
Antifolates
(inhibit bacterial
purine metabolism,
thereby inhibiting
DNA and RNA
synthesis) | | DHFR inhibitor | |
|---|
Sulfonamides
(DHPS inhibitor) | | Short-acting | |
|---|
| Intermediate-acting | |
|---|
| Long-acting | |
|---|
| Other/ungrouped | |
|---|
|
|---|
| Combinations | |
|---|
| Other DHPS inhibitors | |
|---|
|
|---|
Quinolones
(inhibit bacterial
topoisomerase
and/or DNA gyrase,
thereby inhibiting
DNA replication) | | 1st generation | |
|---|
| Fluoroquinolones | | 2nd generation | |
|---|
| 3rd generation | |
|---|
| 4th generation | |
|---|
| Veterinary | |
|---|
|
|---|
| Newer non-fluorinated | |
|---|
| Related (DG) | |
|---|
|
|---|
Anaerobic DNA
inhibitors | | Nitroimidazole derivatives | |
|---|
|
|---|
| RNA synthesis | Rifamycins/
RNA polymerase | |
|---|
| Lipiarmycins | |
|---|
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
Antimycobacterials, including tuberculosis treatment and leprostatic agents (J04) |
|---|
| Nucleic acid inhibitor | Rifamycins/
RNA polymerase inhibitor | |
|---|
| Antifolates/DSI | |
|---|
| ASA | |
|---|
Topoisomerase inhibitors/
quinolones | |
|---|
|
|---|
| Protein synthesis inhibitor | |
|---|
| Cell envelope antibiotic | | Peptidoglycan layer | |
|---|
| Arabinogalactan layer | |
|---|
| Mycolic acid layer |
- Hydrazides/mycolic acid synth. inhibition: Isoniazid#
- Methaniazide
|
|---|
|
|---|
| Other/unknown | |
|---|
| Combinations |
- Ethambutol/isoniazid
- Ethambutol/isoniazid/pyrazinamide/rifampicin#
- Ethambutol/isoniazid/rifampicin#
- Isoniazid/pyridoxine/sulfamethoxazole/trimethoprim#
- Rifampicin/isoniazid/pyrazinamide
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
