Sulfamerazine AHFS/Drugs.com International Drug Names ATC code
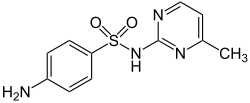
4-amino-N -(4-methylpyrimidin-2-yl)
CAS Number PubChem CID DrugBank ChemSpider UNII KEGG ChEBI ChEMBL CompTox Dashboard (EPA ) ECHA InfoCard 100.004.425 Formula C 11 H 12 N 4 O 2 S Molar mass −1 3D model (JSmol) Melting point 234 to 238 °C (453 to 460 °F)
O=S(=O)(Nc1nc(ccn1)C)c2ccc(N)cc2
InChI=1S/C11H12N4O2S/c1-8-6-7-13-11(14-8)15-18(16,17)10-4-2-9(12)3-5-10/h2-7H,12H2,1H3,(H,13,14,15)
Y Key:QPPBRPIAZZHUNT-UHFFFAOYSA-N
Y (verify)
Sulfamerazine is a sulfonamide antibacterial.
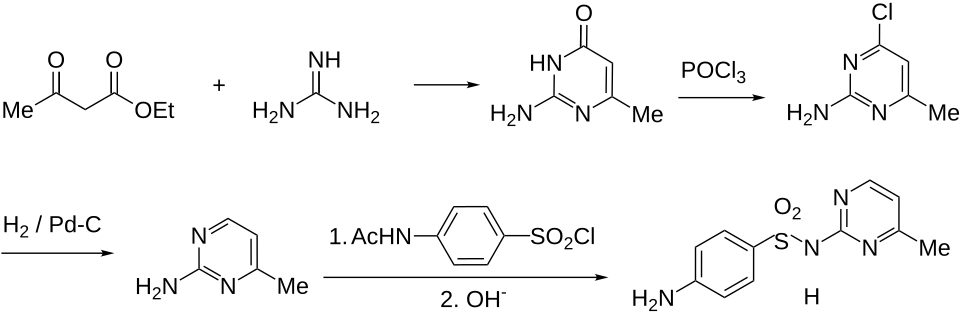
Synthesis
Sulfamerazine synthesis:[ 1] [ 2] U.S. patent 2,378,813
See also
References
^ Roblin RO, Williams JH, Winnek PS, English JP (1940). "Chemotherapy. II. Some Sulfanilamido Heterocycles1". Journal of the American Chemical Society . 62 (8): 2002– 2005. doi:10.1021/ja01865a027 . ^ Sprague JM, Kissinger LW, Lincoln RM (1941). "Sulfonamido Derivatives of Pyrimidines". Journal of the American Chemical Society . 63 (11): 3028– 3030. doi:10.1021/ja01856a046 .
External links
Sulfamerazine at Wikimedia Commons
Antibiotics and chemotherapeutics for dermatological use (D06)
Antibiotics
Tetracycline and derivativesOthers
Streptogramin: Virginiamycin
Chemotherapeutics
Sulfonamides Antivirals Other
Antibacterials that inhibit nucleic acid (J01E, J01M)
Antifolates
DHFR inhibitor Sulfonamides
Short-acting Intermediate-acting Long-acting Other/ungrouped
Combinations Other DHPS inhibitors
Quinolones
1st generation Fluoroquinolones
2nd generation 3rd generation 4th generation Veterinary
Newer non-fluorinated Related (DG)
Anaerobic DNA
Nitroimidazole derivatives
RNA synthesis
Rifamycins/ Lipiarmycins
# WHO-EM‡ Withdrawn from marketClinical trials:
† Phase III§ Never to phase III
![]() Media related to Sulfamerazine at Wikimedia Commons
Media related to Sulfamerazine at Wikimedia Commons