Sulfoxone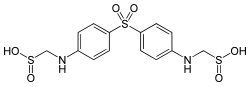 |
 |
|
Routes of
administration | Oral |
|---|
| ATC code | |
|---|
|
| Protein binding | 69% |
|---|
| Metabolism | Hepatic |
|---|
| Elimination half-life | 3 to 8 hours |
|---|
|
[4-[4-(sulfinomethylamino) phenyl] sulfonylphenyl] aminomethanesulfinic acid
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
|
| Formula | C14H16N2Na2O6S3 |
|---|
| Molar mass | 450.45 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
[Na+].[Na+].[O-]S(=O)CNc1ccc(cc1)S(=O)(=O)c2ccc(NCS([O-])=O)cc2
|
InChI=1S/C14H16N2O6S3.2Na/c17-23(18)9-15-11-1-5-13(6-2-11)25(21,22)14-7-3-12(4-8-14)16-10-24(19)20;;/h1-8,15-16H,9-10H2,(H,17,18)(H,19,20);;/q;2*+1/p-2  Y YKey:AZBNFLZFSZDPQF-UHFFFAOYSA-L  Y Y
|
 N N Y (what is this?) (verify) Y (what is this?) (verify) |
Sulfoxone or aldesulfone sodium is an anti-leprosy drug.[1] It is also known as diasone. Sulfoxone sodium was introduced in Japan in 1948.[2] Ernest Muir introduced it to Western use while serving as superintendent of the Chacachacare Leprosarium on Trinidad in the Caribbean.[3]
References
- ^ "Sulfoxone".
- ^ Ozawa H, Maruyama Y (2002). "[A 50-year history of new drugs in Japan: the developments of antileprosy drugs and their epidemiological aspects]". Yakushigaku Zasshi. 37 (1): 76–83. PMID 12412600.
- ^ Browne, Stanley George (1974), "Ernest Muir, C.M.G., C.I.E., M.D. (Edin.), F.R.C.S., LL.D. 1880–1974" (PDF), International Journal of Leprosy, vol. 42, no. 4, Bauru: International Leprosy Association, pp. 457–458, PMID 4617724.
Antimycobacterials, including tuberculosis treatment and leprostatic agents (J04) |
|---|
| Nucleic acid inhibitor | Rifamycins/
RNA polymerase inhibitor | |
|---|
| Antifolates/DSI | |
|---|
| ASA | |
|---|
Topoisomerase inhibitors/
quinolones | |
|---|
|
|---|
| Protein synthesis inhibitor | |
|---|
| Cell envelope antibiotic | | Peptidoglycan layer | |
|---|
| Arabinogalactan layer | |
|---|
| Mycolic acid layer |
- Hydrazides/mycolic acid synth. inhibition: Isoniazid#
- Methaniazide
|
|---|
|
|---|
| Other/unknown | |
|---|
| Combinations |
- Ethambutol/isoniazid
- Ethambutol/isoniazid/pyrazinamide/rifampicin#
- Ethambutol/isoniazid/rifampicin#
- Isoniazid/pyridoxine/sulfamethoxazole/trimethoprim#
- Rifampicin/isoniazid/pyrazinamide
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
Antibacterials that inhibit nucleic acid (J01E, J01M) |
|---|
Antifolates
(inhibit bacterial
purine metabolism,
thereby inhibiting
DNA and RNA
synthesis) | | DHFR inhibitor | |
|---|
Sulfonamides
(DHPS inhibitor) | | Short-acting | |
|---|
| Intermediate-acting | |
|---|
| Long-acting | |
|---|
| Other/ungrouped | |
|---|
|
|---|
| Combinations | |
|---|
| Other DHPS inhibitors | |
|---|
|
|---|
Quinolones
(inhibit bacterial
topoisomerase
and/or DNA gyrase,
thereby inhibiting
DNA replication) | | 1st generation | |
|---|
| Fluoroquinolones | | 2nd generation | |
|---|
| 3rd generation | |
|---|
| 4th generation | |
|---|
| Veterinary | |
|---|
|
|---|
| Newer non-fluorinated | |
|---|
| Related (DG) | |
|---|
|
|---|
Anaerobic DNA
inhibitors | | Nitroimidazole derivatives | |
|---|
|
|---|
| RNA synthesis | Rifamycins/
RNA polymerase | |
|---|
| Lipiarmycins | |
|---|
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|

