Potassium tetrachloropalladate(II)
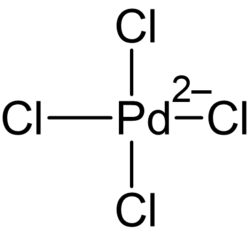
| |
| Names | |
|---|---|
| IUPAC name
dipotassium; tetra chloropalladium(2-)
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.030.033 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
Cl4K2Pd |
| Molar mass | 326.42 g·mol−1 |
| Appearance | dark brown crystals |
| Density | 2.67 g/cm3 |
| Melting point | 525 °C |
Solubility in water
|
soluble |
| Solubility | poorly soluble in ethanol and acetone[1][2] |
| Structure[3] | |
Crystal structure
|
tetragonal |
Space group
|
P 4/mmm |
Lattice constant
|
a = 0.706 nm, c = 0.410 nm
|
Formula units (Z)
|
1 unit per cell |
| Hazards | |
| GHS labelling:[4] | |
Pictograms
|

|
Signal word
|
Warning |
Precautionary statements
|
P261, P305, P338, P351 |
| Related compounds | |
Other cations
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Potassium tetrachloropalladate(II) is an inorganic compound with the chemical formula K2PdCl4.[5][6] It is a dark brown solid, forming tetragonal crystals.
Synthesis
Potassium tetrachloropalladate(II) can be prepared by passing chlorine through a palladium black suspension in a concentrated potassium chloride solution:
- Pd + Cl2 + 2KCl → K2[PdCl4]
It can also be prepared by mixing concentrated solutions of palladium(II) chloride and potassium chloride:[7]
- PdCl2 + 2KCl → K2[PdCl4]
It forms in the decomposition of potassium hexachloropalladate(IV):
- K2[PdCl6] → K2[PdCl4] + Cl2
Uses
The compound is used in the gelation of colloidal semiconductor nanocrystals.[8]
Reactions
The compound reacts with aqua regia forming potassium hexachloropalladate(IV):
- 3K2[PdCl4] + 6HCl + 2HNO3 → 3K2[PdCl6] + 2NO + 4H2O
References
- ^ Perry, Dale L. (19 April 2016). Handbook of Inorganic Compounds. CRC Press. p. 334. ISBN 978-1-4398-1462-8. Retrieved 12 March 2025.
- ^ "Potassium tetrachloropalladate(II), min 32.0% Pd, Thermo Scientific Chemicals, Quantity: 2 g | Fisher Scientific". Fisher Scientific. Retrieved 12 March 2025.
- ^ "K₂PdCl₄". Materials Project. Retrieved 12 March 2025.
- ^ "Potassium tetrachloropalladate(II)". Sigma Aldrich. Retrieved 12 March 2025.
- ^ "Potassium Tetrachloropalladate(II) | AMERICAN ELEMENTS®". American Elements. Retrieved 12 March 2025.
- ^ Bingham, Eula; Cohrssen, Barbara (31 July 2012). Patty's Toxicology, 6 Volume Set. John Wiley & Sons. p. 699. ISBN 978-0-470-41081-3. Retrieved 12 March 2025.
- ^ Basolo, Fred; Burmeister, John L. (2003). On Being Well-coordinated: A Half-century of Research on Transition Metal Complexes : Selected Papers of Fred Basolo. World Scientific. p. 272. ISBN 978-981-238-087-6. Retrieved 12 March 2025.
- ^ Singh, Amita; Lindquist, Beth A.; Ong, Gary K.; Jadrich, Ryan B.; Singh, Ajay; Ha, Heonjoo; Ellison, Christopher J.; Truskett, Thomas M.; Milliron, Delia J. (December 2015). "Linking Semiconductor Nanocrystals into Gel Networks through All‐Inorganic Bridges". Angewandte Chemie International Edition. 54 (49): 14840. doi:10.1002/anie.201508641. ISSN 1433-7851.