Sodium tetrachloropalladate
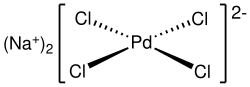 | |
 | |
| Names | |
|---|---|
| IUPAC name
disodium tetrachloropalladium(2+)
| |
| Other names
disodium tetrachloropalladate(II)
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.034.079 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
Cl4Na2Pd |
| Molar mass | 294.20 g·mol−1 |
| Hazards | |
| GHS labelling: | |
Pictograms
|
   
|
Signal word
|
Danger |
Hazard statements
|
H290, H301, H302, H317, H318, H319, H410 |
Precautionary statements
|
P234, P261, P264, P270, P272, P273, P280, P301+P310, P301+P312, P302+P352, P305+P351+P338, P310, P321, P330, P333+P313, P337+P313, P363, P390, P391, P404, P405, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Sodium tetrachloropalladate is an inorganic compound with the chemical formula Na2PdCl4. The compound crystallizes from water as a reddish-brown trihydrate (Na2PdCl4·3H2O), which is the commercially available form.[1]
Preparation
This salt, and the analogous alkali metal salts of the form M2PdCl4, may be prepared simply by reacting palladium(II) chloride with the appropriate alkali metal chloride in aqueous solution.[2] Palladium(II) chloride is insoluble in water, whereas the product dissolves:
- PdCl2 + 2 MCl → M2PdCl4
Related compounds
This compound may further react with phosphines to give phosphine complexes of palladium.
An alternative method of preparing such phosphine complexes is to break up the coordination polymer of palladium(II) chloride into reactive, monomeric acetonitrile or benzonitrile complexes,[3] followed by reaction with phosphines.[2]
References
- ^ The compound's page in Strem Chemicals catalog
- ^ a b Daniele Choueiry and Ei-ichi Negishi (2002). "II.2.3 Pd(0) and Pd(II) Complexes Containing Phosphorus and Other Group 15 Atom Ligands" (Google Books excerpt). In Ei-ichi Negishi (ed.). Handbook of Organopalladium Chemistry for Organic Synthesis. John Wiley & Sons, Inc. ISBN 0-471-31506-0.
- ^ Gordon K. Anderson; Minren Lin (1990). "Bis(Benzonitrile)Dichloro Complexes of Palladium and Platinum". Inorganic Syntheses. Inorganic Syntheses. Vol. 28. pp. 60–63. doi:10.1002/9780470132593.ch13. ISBN 9780470132593.