Sodium bromate
 | |
| Names | |
|---|---|
| IUPAC name
Sodium bromate
| |
| Other names
Sodium bromate(V)
Bromic acid, sodium salt | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.029.237 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1494 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
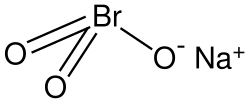
Chemical formula
|
NaBrO3 |
| Molar mass | 150.89g/mol |
| Appearance | colorless or white solid |
| Odor | odorless |
| Density | 3.339 g/cm3 |
| Melting point | 381 °C (718 °F; 654 K) |
| Boiling point | 1,390 °C (2,530 °F; 1,660 K) |
Solubility in water
|
27.5 g/100 mL (0 °C) 36.4 g/100 mL (20 °C) 48.8 g/100 mL (40 °C) 90.8 g/100 mL (100 °C) |
| Solubility | soluble in ammonia insoluble in ethanol |
Magnetic susceptibility (χ)
|
−44.2·10−6 cm3/mol |
Refractive index (nD)
|
1.594 |
| Structure | |
Crystal structure
|
cubic |
| Thermochemistry | |
Std molar
entropy (S⦵298) |
130.5 J/mol K |
Std enthalpy of
formation (ΔfH⦵298) |
−342.5 kJ/mol |
Gibbs free energy (ΔfG⦵)
|
−252.6 kJ/mol |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Oxidizing agent |
| GHS labelling: | |
Pictograms
|
  
|
Signal word
|
Danger |
Hazard statements
|
H271, H272, H302, H315, H319, H335, H341, H350 |
Precautionary statements
|
P201, P202, P210, P220, P221, P261, P264, P270, P271, P280, P281, P283, P301+P312, P302+P352, P304+P340, P305+P351+P338, P306+P360, P308+P313, P312, P321, P330, P332+P313, P337+P313, P362, P370+P378, P371+P380+P375, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 
1
0
0 OX |
| Flash point | 381 °C (718 °F; 654 K) |
| Safety data sheet (SDS) | ICSC 0196 |
| Related compounds | |
Other anions
|
Sodium chlorate Sodium iodate |
Other cations
|
Potassium bromate Calcium bromate |
Related compounds
|
Sodium bromide Sodium hypobromite Sodium bromite |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Sodium bromate, the inorganic compound with the chemical formula of NaBrO3, is the sodium salt of bromic acid. It is a strong oxidant.[1]
Uses
Sodium bromate is mainly used in continuous or batch dyeing processes involving sulfur or vat dyes and as a hair-permagent. In gold mining, it is used in combination with sodium bromide to dissolve gold.
In organic chemistry, sodium bromate is used for a variety of oxidations such as the conversion of alcohols to ketones.[2] Some such oxidations proceed with scission of C-C bonds.[3]
Production
Sodium bromate can be produced from a solution of sodium carbonate and bromine using chlorine gas as the oxidising agent.[4]
- 6 Na2CO3 + Br2 + 5 Cl2 → 2 NaBrO3 + 10 NaCl + 6 CO2
It may also be produced by the electrolytic oxidation of aqueous sodium bromide.[5]
Human health issues
Bromate in drinking water is undesirable because it is a suspected human carcinogen.[6][7] Its presence in Coca-Cola's Dasani bottled water forced a recall of that product in the UK.[8]
References
- ^ Yoffe, David; Frim, Ron; Ukeles, Shmuel D.; Dagani, Michael J.; Barda, Henry J.; Benya, Theodore J.; Sanders, David C. (2013). "Bromine Compounds". Ullmann's Encyclopedia of Industrial Chemistry. pp. 1–31. doi:10.1002/14356007.a04_405.pub2. ISBN 978-3-527-30385-4.
- ^ Harrison, James J.; Kulkarni, Amol A. (2013). "Sodium Bromate". Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rs053.pub2. ISBN 978-0-471-93623-7.
- ^ Donald A. Ballard, William M. Dehn (1921). "Benzilic Acid". Organic Syntheses. 1: 29. doi:10.15227/orgsyn.001.0029.
- ^ CN patent 1034653C, Zhao Naizhi; Wu Zongsheng; Sun Guishi, published 1997-04-23, assigned to Daqinghe Saltern Changlu Hebei Prov.
- ^ WO patent 1996030562A1, David B. Blum; Rodney H. Sergent; Vadim Zolotarsky, "Process and apparatus for generating bromine", published 1996-10-03, assigned to Electrocatalytic Inc
- ^ "Potassium Bromate (Group 2B)". International Agency for Research on Cancer: Summaries and Evaluations. Canadian Centre for Occupational Health and Safety. Retrieved 2008-03-09.
- ^ Kurokawa, Yuji; Maekawa, A; Takahashi, M; Hayashi, Y (July 1990). "Toxicity and carcinogenicity of potassium bromate—a new renal carcinogen". Environmental Health Perspectives. 87: 309–35. doi:10.1289/EHP.9087309. JSTOR 3431039. PMC 1567851. PMID 2269236.
- ^ "Coke recalls controversial water". BBC News. 2004-03-19. Retrieved 2008-03-09.
Notes
External links