Metharbital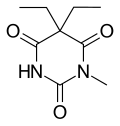 |
 |
|
| Other names | Endiemal, metharbitone, methobarbitone[1] |
|---|
Routes of
administration | By mouth (tablets) |
|---|
| ATC code | |
|---|
|
| Legal status |
- CA: Schedule IV
- US: Schedule III
|
|---|
|
5,5-Diethyl-1-methylpyrimidine-2,4,6(1H,3H,5H)-trione
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| IUPHAR/BPS | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
| ECHA InfoCard | 100.000.011 |
|---|
|
| Formula | C9H14N2O3 |
|---|
| Molar mass | 198.222 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
O=C1N(C(=O)NC(=O)C1(CC)CC)C
|
InChI=1S/C9H14N2O3/c1-4-9(5-2)6(12)10-8(14)11(3)7(9)13/h4-5H2,1-3H3,(H,10,12,14)  Y YKey:FWJKNZONDWOGMI-UHFFFAOYSA-N  Y Y
|
| (verify) |
Metharbital was patented in 1905 by Emil Fischer working for Merck.[2] It was marketed as Gemonil by Abbott Laboratories. It is a barbiturate anticonvulsant, used in the treatment of epilepsy.[3][4] It has similar properties to phenobarbital.
History
- 1952 Gemonil was introduced by Abbott Laboratories.
- 1990 Abbott stopped marketing.
Synthesis
Metharbital can be synthesized from 2,2-diethylmalonic acid and O-methylisourea.[5][6][2]
References
- ^ "Metharbital". The Comparative Toxicogenomics Database.
- ^ a b US 782742, Fischer E, "Trisubstituted barbituric acids and process of making them.", issued 14 February 1905, assigned to E. Merck
- ^ Shorvon SR, Fish DR, Perucca E, Dodson WE, eds. (2004). The Treatment of Epilepsy (2nd ed.). Blackwell. ISBN 0-632-06046-8.
- ^ Resor SR (1991). The Medical Treatment of Epilepsy. Marcel Dekker. ISBN 0-8247-8549-5.
- ^ Halpern A, Jones JW (June 1949). "The characterization of the trialkylbarbiturates". Journal of the American Pharmaceutical Association. 38 (6): 352–5. doi:10.1002/jps.3030380619. PMID 18151714.
- ^ Snyder JA, Link KP (1953). "Preparation and Characterization by Alkaline Methanolysis of 5,5-Diethyl-4-(tetraacetyl-β-D-glucosyloxy)-2,6(1,5)-pyrimidinedione". Journal of the American Chemical Society. 75 (8): 1881–1883. doi:10.1021/ja01104a030.
Anticonvulsants (N03) |
|---|
| GABAergics | | GABAAR PAMs | |
|---|
| GABA-T inhibitors | |
|---|
| Others | |
|---|
|
|---|
Channel
modulators | | Sodium blockers | |
|---|
| Calcium blockers | |
|---|
| Potassium openers | |
|---|
|
|---|
| Others | |
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
GABAA receptor positive modulators |
|---|
| Alcohols | |
|---|
| Barbiturates | |
|---|
| Benzodiazepines | |
|---|
| Carbamates |
- Carisbamate
- Carisoprodol
- Clocental
- Cyclarbamate
- Difebarbamate
- Emylcamate
- Ethinamate
- Febarbamate
- Felbamate
- Hexapropymate
- Hydroxyphenamate
- Lorbamate
- Mebutamate
- Meprobamate
- Nisobamate
- Pentabamate
- Phenprobamate
- Procymate
- Styramate
- Tetrabamate
- Tybamate
|
|---|
| Flavonoids | |
|---|
| Imidazoles |
- Etomidate
- Metomidate
- Methoxyetomidate
- Propoxate
- Isopropoxate
- Butomidate
- Iso-butomidate
- Sec-butomidate
- CF2-Etomidate
- CF3-Etomidate
- CF3-Propoxate
- Flutomidate
- 2,6-Dichloro-3-fluoroetomidate
|
|---|
| Kava constituents |
- 10-Methoxyyangonin
- 11-Methoxyyangonin
- 11-Hydroxyyangonin
- Desmethoxyyangonin
- 11-Methoxy-12-hydroxydehydrokavain
- 7,8-Dihydroyangonin
- Kavain
- 5-Hydroxykavain
- 5,6-Dihydroyangonin
- 7,8-Dihydrokavain
- 5,6,7,8-Tetrahydroyangonin
- 5,6-Dehydromethysticin
- Methysticin
- 7,8-Dihydromethysticin
- Yangonin
|
|---|
| Monoureides |
- Acecarbromal
- Apronal (apronalide)
- Bromisoval
- Carbromal
- Capuride
- Ectylurea
|
|---|
| Neuroactive steroids | |
|---|
| Nonbenzodiazepines |
- Imidazopyridines: Alpidem
- DS-1
- Necopidem
- Saripidem
- Zolpidem
- Pyrazolopyrimidines: Divaplon
- Fasiplon
- Indiplon
- Lorediplon
- Ocinaplon
- Panadiplon
- Taniplon
- Zaleplon
- Others: Adipiplon
- AXS-17 (BAER-101, AZD-7325)
- CGS-8216
- CGS-9896
- CGS-13767
- CGS-20625
- CL-218,872
- CP-615,003
- CTP-354
- ELB-139
- GBLD-345
- Imepitoin
- JM-1232
- L-838,417
- Lirequinil (Ro41-3696)
- Miltirone (rosmariquinone)
- NS-2664
- NS-2710
- NS-11394
- Pipequaline
- ROD-188
- RWJ-51204
- SB-205,384
- SX-3228
- TGSC01AA
- TP-003
- TPA-023
- TP-13
- U-89843A
- U-90042
- Viqualine
- Y-23684
|
|---|
| Phenols | |
|---|
| Piperidinediones | |
|---|
| Pyrazolopyridines |
- Cartazolate
- Etazolate
- ICI-190,622
- Tracazolate
|
|---|
| Quinazolinones |
- Afloqualone
- Cloroqualone
- Diproqualone
- Etaqualone
- Mebroqualone
- Mecloqualone
- Methaqualone
- Methylmethaqualone
- Nitromethaqualone
- SL-164
|
|---|
| Volatiles/gases | |
|---|
| Others/unsorted |
- 3-Hydroxybutanal
- α-EMTBL
- AA-29504
- Alogabat
- Avermectins (e.g., ivermectin)
- Bromide compounds (e.g., lithium bromide, potassium bromide, sodium bromide)
- Carbamazepine
- Chloralose
- Chlormezanone
- Clomethiazole
- Darigabat
- DEABL
- Deuterated etifoxine
- Dihydroergolines (e.g., dihydroergocryptine, dihydroergosine, dihydroergotamine, ergoloid (dihydroergotoxine))
- DS2
- Efavirenz
- Etazepine
- Etifoxine
- Fenamates (e.g., flufenamic acid, mefenamic acid, niflumic acid, tolfenamic acid)
- Fluoxetine
- Flupirtine
- Hopantenic acid
- KRM-II-81
- Lanthanum
- Lavender oil
- Lignans (e.g., 4-O-methylhonokiol, honokiol, magnolol, obovatol)
- Loreclezole
- Menthyl isovalerate (validolum)
- Monastrol
- Nicotinic acid
- Nicotinamide
- Org 25,435
- Phenytoin
- Propanidid
- Retigabine (ezogabine)
- Safranal
- Seproxetine
- Stiripentol
- Sulfonylalkanes (e.g., sulfonmethane (sulfonal), tetronal, trional)
- Terpenoids (e.g., borneol)
- Topiramate
- Valerian constituents (e.g., isovaleric acid, isovaleramide, valerenic acid, valerenol)
- Unsorted benzodiazepine site positive modulators: α-Pinene
- MRK-409 (MK-0343)
- TCS-1105
- TCS-1205
|
|---|
See also: Receptor/signaling modulators • GABA receptor modulators • GABA metabolism/transport modulators |

