Fludiazepam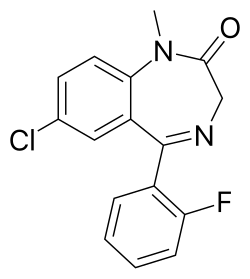 |
 |
|
| Trade names | Erispan (JP, TW) |
|---|
| AHFS/Drugs.com | International Drug Names |
|---|
Routes of
administration | Oral (tablets) |
|---|
| Drug class | Benzodiazepine |
|---|
| ATC code | |
|---|
|
| Legal status |
- BR: Class B1 (Psychoactive drugs)[1]
- CA: Schedule IV
- DE: Anlage III (Special prescription form required)
- US: Schedule IV
|
|---|
|
| Metabolism | Hepatic |
|---|
| Excretion | Renal |
|---|
|
7-chloro-5-(2-fluorophenyl)-1-methyl-1,3-dihydro-2H-1,4-benzodiazepin-2-one
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEBI | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
| ECHA InfoCard | 100.292.343 |
|---|
|
| Formula | C16H12ClFN2O |
|---|
| Molar mass | 302.73 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
O=C1CN=C(C2=CC=CC=C2F)C3=CC(Cl)=CC=C3N1C
|
InChI=1S/C16H12ClFN2O/c1-20-14-7-6-10(17)8-12(14)16(19-9-15(20)21)11-4-2-3-5-13(11)18/h2-8H,9H2,1H3  Y YKey:ROYOYTLGDLIGBX-UHFFFAOYSA-N  Y Y
|
| (verify) |
Fludiazepam,[2] marketed under the brand name Erispan (エリスパン)[3][4] is a potent benzodiazepine and 2ʹ-fluoro derivative of diazepam,[5] originally developed by Hoffmann-La Roche in the 1960s.[6] It is marketed in Japan and Taiwan. It exerts its pharmacological properties via enhancement of GABAergic inhibition.[7] Fludiazepam has 4 times more binding affinity for benzodiazepine receptors than diazepam.[8] It possesses anxiolytic,[9][10][11] anticonvulsant, sedative, hypnotic and skeletal muscle relaxant properties.[12] Fludiazepam has been used recreationally.[13]
See also
- Diazepam
- Diclazepam (the 2ʹ-chloro analog)
- Difludiazepam (the 2',6'-difluoro derivative)
- Flunitrazepam (the 7-nitro analog)
- Flualprazolam (the triazolo derivative)
- Ro20-8552
References
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ US Patent 3371085 5-aryl-3h-1,4-benzodiazepin-2(1h)-ones
- ^ Su TP, Chen TJ, Hwang SJ, Chou LF, Fan AP, Chen YC (August 2002). "Utilization of psychotropic drugs in Taiwan: an overview of outpatient sector in 2000". Zhonghua Yi Xue Za Zhi (Chinese Medical Journal; Free China Ed). 65 (8): 378–91. PMID 12455808.
- ^ "Benzodiazepine Names". non-benzodiazepines.org.uk. Archived from the original on 2008-12-08. Retrieved 2008-12-29.
- ^ Neville GA, Beckstead HD, Shurvell HF (February 1994). "A Fourier transform-Raman and infrared vibrational study of delorazepam, fludiazepam, flurazepam, and tetrazepam". Journal of Pharmaceutical Sciences. 83 (2): 143–51. doi:10.1002/jps.2600830207. PMID 7909552.
- ^ US Patent 3299053 -ARYL-JH-L,X-BENZODIAZEPIN-Z(LH)-ONES
- ^ Tsuchiya T, Fukushima H (April 1978). "Effects of benzodiazepines and pentobarbitone on the gaba-ergic recurrent inhibition of hippocampal neurons". European Journal of Pharmacology. 48 (4): 421–4. doi:10.1016/0014-2999(78)90169-3. PMID 648585.
- ^ Nakatsuka I, Shimizu H, Asami Y, Katoh T, Hirose A, Yoshitake A (January 1985). "Benzodiazepines and their metabolites: relationship between binding affinity to the benzodiazepine receptor and pharmacological activity". Life Sciences. 36 (2): 113–9. doi:10.1016/0024-3205(85)90089-X. PMID 2857046.
- ^ Okada S, Ichiki K, Tanokuchi S, Ishii K, Hamada H, Ota Z (1994). "Effect of an anxiolytic on lipid profile in non-insulin-dependent diabetes mellitus". The Journal of International Medical Research. 22 (6): 338–42. doi:10.1177/030006059402200605. PMID 7895897. S2CID 9591519.
- ^ Okada S, Ichiki K, Tanokuchi S, Ishii K, Hamada H, Ota Z (1995). "Improvement of stress reduces glycosylated haemoglobin levels in patients with type 2 diabetes". The Journal of International Medical Research. 23 (2): 119–22. doi:10.1177/030006059502300205. PMID 7601294. S2CID 12865838.
- ^ Okada S, Ichiki K, Tanokuchi S, Ishii K, Hamada H, Ota Z (1995). "How blood pressure in patients with non-insulin-dependent diabetes mellitus is influenced by stress". The Journal of International Medical Research. 23 (5): 377–80. doi:10.1177/030006059502300508. PMID 8529781. S2CID 42485678.
- ^ Inoue H, Maeno Y, Iwasa M, Matoba R, Nagao M (September 2000). "Screening and determination of benzodiazepines in whole blood using solid-phase extraction and gas chromatography/mass spectrometry". Forensic Science International. 113 (1–3): 367–73. doi:10.1016/S0379-0738(00)00226-7. PMID 10978650.
- ^ Shimamine M, Masunari T, Nakahara Y (1993). "[Studies on identification of drugs of abuse by diode array detection. I. Screening-test and identification of benzodiazepines by HPLC-DAD with ICOS software system]". Eisei Shikenjo Hokoku. Bulletin of National Institute of Hygienic Sciences (111): 47–56. PMID 7920567.
External links
Benzodiazepines |
|---|
| 1,4-Benzodiazepines | |
|---|
| 1,5-Benzodiazepines |
- Arfendazam
- Clobazam
- CP-1414S
- Lofendazam
- Triflubazam
|
|---|
| 2,3-Benzodiazepines* |
- Girisopam
- GYKI-52466
- GYKI-52895
- Nerisopam
- Talampanel
- Tofisopam
|
|---|
| Triazolobenzodiazepines |
- Adinazolam
- Alprazolam
- Balovaptan*
- Bromazolam
- Clonazolam
- Estazolam
- Fluadinazolam
- Flualprazolam
- Flubromazolam
- Flunitrazolam
- Nitrazolam
- Phenazolam
- Pyrazolam
- Rilmazolam (active metabolite of Rilmazafone)
- Triazolam
|
|---|
| Imidazobenzodiazepines |
- Bretazenil
- Climazolam
- EVT-201
- FG-8205
- Flumazenil
- GL-II-73
- Imidazenil
- 123I-Iomazenil
- L-655,708
- Loprazolam
- Midazolam
- PWZ-029
- Remimazolam
- Ro15-4513
- Ro48-6791
- Ro48-8684
- Ro4938581
- Sarmazenil
- SH-053-R-CH3-2′F
|
|---|
| Oxazolobenzodiazepines | |
|---|
| Thienodiazepines | |
|---|
| Thienotriazolodiazepines |
- α-Hydroxyetizolam
- Apafant*
- Brotizolam
- Ciclotizolam
- Clotizolam
- Deschloroclotizolam
- Deschloroetizolam
- Etizolam
- Flubrotizolam
- Fluclotizolam
- Fluetizolam
- Israpafant*
- JQ1*
- Metizolam
|
|---|
| Thienobenzodiazepines* | |
|---|
| Pyridodiazepines | |
|---|
| Pyridotriazolodiazepines | |
|---|
| Pyrazolodiazepines |
- Razobazam*
- Ripazepam
- Zolazepam
- Zomebazam
- Zometapine*
|
|---|
| Pyrrolodiazepines | |
|---|
| Tetrahydroisoquinobenzodiazepines | |
|---|
| Pyrrolobenzodiazepines* | |
|---|
| Benzodiazepine prodrugs |
- Alprazolam triazolobenzophenone
- Avizafone
- Rilmazafone
|
|---|
* atypical activity profile (not GABAA receptor ligands) |
Anxiolytics (N05B) |
|---|
| 5-HT1ARTooltip 5-HT1A receptor agonists | |
|---|
| GABAARTooltip GABAA receptor PAMsTooltip positive allosteric modulators | |
|---|
Gabapentinoids
(α2δ VDCC blockers) | |
|---|
| Antidepressants |
- SSRIsTooltip Selective serotonin reuptake inhibitors (e.g., escitalopram)
- SNRIsTooltip Serotonin-norepinephrine reuptake inhibitors (e.g., duloxetine)
- SARIsTooltip Serotonin antagonist and reuptake inhibitors (e.g., trazodone)
- TCAsTooltip Tricyclic antidepressants (e.g., clomipramine#)
- TeCAsTooltip Tetracyclic antidepressants (e.g., mirtazapine)
- MAOIsTooltip Monoamine oxidase inhibitors (e.g., phenelzine); Others: Agomelatine
- Bupropion
- Tianeptine
- Vilazodone
- Vortioxetine
|
|---|
Sympatholytics
(Antiadrenergics) |
- Alpha-1 blockers (e.g., prazosin)
- Alpha-2 agonists (e.g., clonidine, dexmedetomidine, guanfacine)
- Beta blockers (e.g., propranolol, atenolol, betaxolol, nadolol, oxprenolol, pindolol)
|
|---|
| Others | |
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
GABAA receptor positive modulators |
|---|
| Alcohols | |
|---|
| Barbiturates | |
|---|
| Benzodiazepines | |
|---|
| Carbamates |
- Carisbamate
- Carisoprodol
- Clocental
- Cyclarbamate
- Difebarbamate
- Emylcamate
- Ethinamate
- Febarbamate
- Felbamate
- Hexapropymate
- Hydroxyphenamate
- Lorbamate
- Mebutamate
- Meprobamate
- Nisobamate
- Pentabamate
- Phenprobamate
- Procymate
- Styramate
- Tetrabamate
- Tybamate
|
|---|
| Flavonoids | |
|---|
| Imidazoles |
- Etomidate
- Metomidate
- Methoxyetomidate
- Propoxate
- Isopropoxate
- Butomidate
- Iso-butomidate
- Sec-butomidate
- CF2-Etomidate
- CF3-Etomidate
- CF3-Propoxate
- Flutomidate
- 2,6-Dichloro-3-fluoroetomidate
|
|---|
| Kava constituents |
- 10-Methoxyyangonin
- 11-Methoxyyangonin
- 11-Hydroxyyangonin
- Desmethoxyyangonin
- 11-Methoxy-12-hydroxydehydrokavain
- 7,8-Dihydroyangonin
- Kavain
- 5-Hydroxykavain
- 5,6-Dihydroyangonin
- 7,8-Dihydrokavain
- 5,6,7,8-Tetrahydroyangonin
- 5,6-Dehydromethysticin
- Methysticin
- 7,8-Dihydromethysticin
- Yangonin
|
|---|
| Monoureides |
- Acecarbromal
- Apronal (apronalide)
- Bromisoval
- Carbromal
- Capuride
- Ectylurea
|
|---|
| Neuroactive steroids | |
|---|
| Nonbenzodiazepines |
- Imidazopyridines: Alpidem
- DS-1
- Necopidem
- Saripidem
- Zolpidem
- Pyrazolopyrimidines: Divaplon
- Fasiplon
- Indiplon
- Lorediplon
- Ocinaplon
- Panadiplon
- Taniplon
- Zaleplon
- Others: Adipiplon
- AXS-17 (BAER-101, AZD-7325)
- CGS-8216
- CGS-9896
- CGS-13767
- CGS-20625
- CL-218,872
- CP-615,003
- CTP-354
- ELB-139
- GBLD-345
- Imepitoin
- JM-1232
- L-838,417
- Lirequinil (Ro41-3696)
- Miltirone (rosmariquinone)
- NS-2664
- NS-2710
- NS-11394
- Pipequaline
- ROD-188
- RWJ-51204
- SB-205,384
- SX-3228
- TGSC01AA
- TP-003
- TPA-023
- TP-13
- U-89843A
- U-90042
- Viqualine
- Y-23684
|
|---|
| Phenols | |
|---|
| Piperidinediones | |
|---|
| Pyrazolopyridines |
- Cartazolate
- Etazolate
- ICI-190,622
- Tracazolate
|
|---|
| Quinazolinones |
- Afloqualone
- Cloroqualone
- Diproqualone
- Etaqualone
- Mebroqualone
- Mecloqualone
- Methaqualone
- Methylmethaqualone
- Nitromethaqualone
- SL-164
|
|---|
| Volatiles/gases | |
|---|
| Others/unsorted |
- 3-Hydroxybutanal
- α-EMTBL
- AA-29504
- Alogabat
- Avermectins (e.g., ivermectin)
- Bromide compounds (e.g., lithium bromide, potassium bromide, sodium bromide)
- Carbamazepine
- Chloralose
- Chlormezanone
- Clomethiazole
- Darigabat
- DEABL
- Deuterated etifoxine
- Dihydroergolines (e.g., dihydroergocryptine, dihydroergosine, dihydroergotamine, ergoloid (dihydroergotoxine))
- DS2
- Efavirenz
- Etazepine
- Etifoxine
- Fenamates (e.g., flufenamic acid, mefenamic acid, niflumic acid, tolfenamic acid)
- Fluoxetine
- Flupirtine
- Hopantenic acid
- KRM-II-81
- Lanthanum
- Lavender oil
- Lignans (e.g., 4-O-methylhonokiol, honokiol, magnolol, obovatol)
- Loreclezole
- Menthyl isovalerate (validolum)
- Monastrol
- Nicotinic acid
- Nicotinamide
- Org 25,435
- Phenytoin
- Propanidid
- Retigabine (ezogabine)
- Safranal
- Seproxetine
- Stiripentol
- Sulfonylalkanes (e.g., sulfonmethane (sulfonal), tetronal, trional)
- Terpenoids (e.g., borneol)
- Topiramate
- Valerian constituents (e.g., isovaleric acid, isovaleramide, valerenic acid, valerenol)
- Unsorted benzodiazepine site positive modulators: α-Pinene
- MRK-409 (MK-0343)
- TCS-1105
- TCS-1205
|
|---|
See also: Receptor/signaling modulators • GABA receptor modulators • GABA metabolism/transport modulators |

