Fotemustine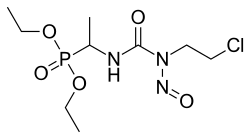 |
|
| Trade names | Mustophoran |
|---|
| AHFS/Drugs.com | International Drug Names |
|---|
Pregnancy
category | |
|---|
| ATC code | |
|---|
|
| Legal status |
- AU: S4 (Prescription only)
|
|---|
|
(RS)-diethyl (1-{[(2-chloroethyl)(nitroso)carbamoyl]amino}
ethyl)phosphonate
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
| ECHA InfoCard | 100.158.792 |
|---|
|
| Formula | C9H19ClN3O5P |
|---|
| Molar mass | 315.69 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
O=P(OCC)(OCC)C(NC(=O)N(N=O)CCCl)C
|
InChI=1S/C9H19ClN3O5P/c1-4-17-19(16,18-5-2)8(3)11-9(14)13(12-15)7-6-10/h8H,4-7H2,1-3H3,(H,11,14)  Y YKey:YAKWPXVTIGTRJH-UHFFFAOYSA-N  Y Y
|
 N N Y (what is this?) (verify) Y (what is this?) (verify) |
Fotemustine is a nitrosourea alkylating agent used in the treatment of metastatic melanoma. It is available in Europe but has not been approved by the United States FDA.[1] A study[2] has shown that fotemustine produces improved response rates but does not increase survival (over dacarbazine in the treatment of disseminated cutaneous melanoma. Median survival was 7.3 months with fotemustine versus 5.6 months with DTIC (P=.067). There was also toxicity prevalence in fotemustine arm. The main toxicity was grade 3 to 4 neutropenia (51% with fotemustine v 5% with DTIC) and thrombocytopenia (43% v 6%, respectively).
External links
Intracellular chemotherapeutic agents / antineoplastic agents (L01) |
|---|
SPs/MIs
(M phase) | | Block microtubule assembly | |
|---|
| Block microtubule disassembly |
- Taxanes (Cabazitaxel
- Docetaxel#
- Larotaxel
- Ortataxel†
- Paclitaxel#
- Tesetaxel)
- Epothilones (Ixabepilone)
|
|---|
|
|---|
DNA replication
inhibitor | DNA precursors/
antimetabolites
(S phase) | | Folic acid | |
|---|
| Purine | |
|---|
| Pyrimidine |
- DNA polymerase inhibitor (Cytarabine# +daunorubicin)
|
|---|
| Deoxyribonucleotide | |
|---|
|
|---|
Topoisomerase inhibitors
(S phase) | | I |
- Camptotheca (Belotecan
- Camptothecin
- Cositecan†
- Etirinotecan pegol†
- Exatecan
- Gimatecan
- Irinotecan#
- Lurtotecan‡
- Rubitecan‡
- Silatecan§
- Topotecan)
|
|---|
| II | |
|---|
| II+Intercalation | |
|---|
|
|---|
Crosslinking of DNA
(CCNS) | | Alkylating | |
|---|
| Platinum-based | |
|---|
| Nonclassical | |
|---|
| Intercalation | |
|---|
|
|---|
|
|---|
| Photosensitizers/PDT | |
|---|
| Other | | Enzyme inhibitors |
- FI (Tipifarnib§)
- CDK inhibitors (Abemaciclib
- Alvocidib†
- Palbociclib
- Ribociclib
- Seliciclib†)
- PrI
- PhI (Anagrelide)
- IMPDI (Tiazofurin§)
- LI (Masoprocol)
- PARP inhibitor (Fuzuloparib
- Niraparib +abiraterone acetate
- Olaparib
- Rucaparib)
- HDAC (Belinostat
- Entinostat
- Panobinostat
- Romidepsin
- Vorinostat)
- PIKI (Pi3K) (Alpelisib
- Copanlisib‡
- Duvelisib
- Idelalisib
- Inavolisib
- Umbralisib‡)
- IDH (Enasidenib
- Ivosidenib
- Olutasidenib
- Vorasidenib)
|
|---|
| Receptor antagonists | |
|---|
| Other/ungrouped |
- Adagrasib
- Aflibercept
- Arsenic trioxide# (Realgar/Indigo naturalis#)
- Asparagine depleters (Asparaginase#/Pegaspargase)
- Axicabtagene ciloleucel
- Belzutifan
- Bexarotene
- Brexucabtagene autoleucel
- Calaspargase pegol
- Celecoxib
- Ciltacabtagene autoleucel
- Darinaparsin
- Demecolcine
- Denileukin diftitox
- Eflornithine
- Elesclomol§
- Elsamitrucin
- Epacadostat
- Eribulin
- Estramustine
- Glasdegib
- Idecabtagene vicleucel
- Idroxioleic acid
- Imetelstat
- Lifileucel
- Lisocabtagene maraleucel
- Lonidamine
- Lucanthone
- Lurbinectedin
- Mitoguazone
- Mitotane
- Nadofaragene firadenovec
- Navitoclax
- Nirogacestat
- Obecabtagene autoleucel
- Oblimersen†
- Omacetaxine mepesuccinate
- Para-toluenesulfonamide
- Pelabresib
- Plitidepsin
- Retinoids (Alitretinoin
- Tretinoin#)
- Selinexor
- Sitimagene ceradenovec
- Sotorasib
- Tabelecleucel
- Tagraxofusp
- Talimogene laherparepvec
- Tazemetostat
- Tebentafusp
- Tiazofurine
- Tigilanol tiglate
- Tisagenlecleucel
- Trabectedin
- Veliparib
- Venetoclax
- Verdinexor
- Vosaroxin
|
|---|
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
