Porfimer sodium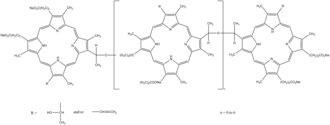 |
|
| Trade names | Photofrin |
|---|
| AHFS/Drugs.com | Consumer Drug Information |
|---|
| License data |
|
|---|
Routes of
administration | Intravenous |
|---|
| ATC code | |
|---|
|
| Legal status |
|
|---|
|
| Bioavailability | NA |
|---|
| Protein binding | ~90% |
|---|
| Elimination half-life | 21.5 days (mean) |
|---|
| Excretion | Fecal |
|---|
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| ChEMBL | |
|---|
|
| Formula | C68H74N8O11 (for n=0) |
|---|
| Molar mass | 1179.36 g/mol (for n=0) g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
[Na+].CC(O)C1=C(C)C=2C=C5NC(=CC4=NC(=CC=3NC(C=C1N=2)=C(C)C=3CCC(O)=O)C(CCC(O)=O)=C4C)C(C)=C5C(C)OC(C)C6=C(C)C=7C=C%10NC(=CC9=NC(=CC=8NC(C=C6N=7)=C(C)C=8CCC(O)=O)C(CCC(O)=O)=C9C)C(C)=C%10C(C)O
|
InChI=1S/C68H74N8O11.Na/c1-29-41(13-17-61(79)80)53-28-56-44(16-20-64(85)86)32(4)48(72-56)24-59-68(36(8)52(76-59)25-58-65(37(9)77)33(5)49(73-58)21-45(29)69-53)40(12)87-39(11)67-35(7)50-22-46-30(2)42(14-18-62(81)82)54(70-46)27-55-43(15-19-63(83)84)31(3)47(71-55)23-57-66(38(10)78)34(6)51(74-57)26-60(67)75-50;/h21-28,37-40,71-73,75,77-78H,13-20H2,1-12H3,(H,79,80)(H,81,82)(H,83,84)(H,85,86);/q;+1/b45-21-,46-22-,47-23-,48-24-,49-21-,50-22-,51-26-,52-25-,53-28-,54-27-,55-27-,56-28-,57-23-,58-25-,59-24-,60-26-;  Y YKey:CGQHMICGJYKFFJ-ZLJVSRBASA-N  Y Y
|
 N N Y (what is this?) (verify) Y (what is this?) (verify) |
Porfimer sodium, sold as Photofrin, is a photosensitizer used in photodynamic therapy and radiation therapy and for palliative treatment of obstructing endobronchial non-small cell lung carcinoma and obstructing esophageal cancer.
Porfimer is a mixture of oligomers formed by ether and ester linkages of up to eight porphyrin units.[1] In practice, a red light source emitting at 630 nm is used to excite the Porfimer oligomers.[2]
Porfimer is Haematoporphyrin Derivative (HpD) (See PDT).
Approvals and indications
It was approved in Canada in 1993 for the treatment of bladder cancer.[2]
It was approved in Japan in 1994 (for early stage lung cancer?).[2]
It was approved by the U.S. FDA in December 1995 for esophageal cancer, and in 1998, it was approved for the treatment of early non-small cell lung cancer.[2]
In August 2003 the FDA approved its use for Barrett's esophagus.[3]
References
External links
Intracellular chemotherapeutic agents / antineoplastic agents (L01) |
|---|
SPs/MIs
(M phase) | | Block microtubule assembly | |
|---|
| Block microtubule disassembly |
- Taxanes (Cabazitaxel
- Docetaxel#
- Larotaxel
- Ortataxel†
- Paclitaxel#
- Tesetaxel)
- Epothilones (Ixabepilone)
|
|---|
|
|---|
DNA replication
inhibitor | DNA precursors/
antimetabolites
(S phase) | | Folic acid | |
|---|
| Purine | |
|---|
| Pyrimidine |
- DNA polymerase inhibitor (Cytarabine# +daunorubicin)
|
|---|
| Deoxyribonucleotide | |
|---|
|
|---|
Topoisomerase inhibitors
(S phase) | | I |
- Camptotheca (Belotecan
- Camptothecin
- Cositecan†
- Etirinotecan pegol†
- Exatecan
- Gimatecan
- Irinotecan#
- Lurtotecan‡
- Rubitecan‡
- Silatecan§
- Topotecan)
|
|---|
| II | |
|---|
| II+Intercalation | |
|---|
|
|---|
Crosslinking of DNA
(CCNS) | | Alkylating | |
|---|
| Platinum-based | |
|---|
| Nonclassical | |
|---|
| Intercalation | |
|---|
|
|---|
|
|---|
| Photosensitizers/PDT | |
|---|
| Other | | Enzyme inhibitors |
- FI (Tipifarnib§)
- CDK inhibitors (Abemaciclib
- Alvocidib†
- Palbociclib
- Ribociclib
- Seliciclib†)
- PrI
- PhI (Anagrelide)
- IMPDI (Tiazofurin§)
- LI (Masoprocol)
- PARP inhibitor (Fuzuloparib
- Niraparib +abiraterone acetate
- Olaparib
- Rucaparib)
- HDAC (Belinostat
- Entinostat
- Panobinostat
- Romidepsin
- Vorinostat)
- PIKI (Pi3K) (Alpelisib
- Copanlisib‡
- Duvelisib
- Idelalisib
- Inavolisib
- Umbralisib‡)
- IDH (Enasidenib
- Ivosidenib
- Olutasidenib
- Vorasidenib)
|
|---|
| Receptor antagonists | |
|---|
| Other/ungrouped |
- Adagrasib
- Aflibercept
- Arsenic trioxide# (Realgar/Indigo naturalis#)
- Asparagine depleters (Asparaginase#/Pegaspargase)
- Axicabtagene ciloleucel
- Belzutifan
- Bexarotene
- Brexucabtagene autoleucel
- Calaspargase pegol
- Celecoxib
- Ciltacabtagene autoleucel
- Darinaparsin
- Demecolcine
- Denileukin diftitox
- Eflornithine
- Elesclomol§
- Elsamitrucin
- Epacadostat
- Eribulin
- Estramustine
- Glasdegib
- Idecabtagene vicleucel
- Idroxioleic acid
- Imetelstat
- Lifileucel
- Lisocabtagene maraleucel
- Lonidamine
- Lucanthone
- Lurbinectedin
- Mitoguazone
- Mitotane
- Nadofaragene firadenovec
- Navitoclax
- Nirogacestat
- Obecabtagene autoleucel
- Oblimersen†
- Omacetaxine mepesuccinate
- Para-toluenesulfonamide
- Pelabresib
- Plitidepsin
- Retinoids (Alitretinoin
- Tretinoin#)
- Selinexor
- Sitimagene ceradenovec
- Sotorasib
- Tabelecleucel
- Tagraxofusp
- Talimogene laherparepvec
- Tazemetostat
- Tebentafusp
- Tiazofurine
- Tigilanol tiglate
- Tisagenlecleucel
- Trabectedin
- Veliparib
- Venetoclax
- Verdinexor
- Vosaroxin
|
|---|
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
