Vindesine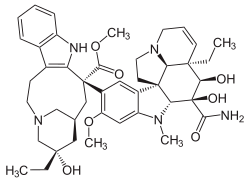 |
 |
|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
|---|
Pregnancy
category | |
|---|
Routes of
administration | Intravenous |
|---|
| ATC code | |
|---|
|
| Legal status |
- In general: ℞ (Prescription only)
|
|---|
|
| Protein binding | 65-75% |
|---|
| Metabolism | Hepatic (CYP3A4-mediated) |
|---|
| Elimination half-life | 24 hours |
|---|
| Excretion | Biliary and renal |
|---|
|
methyl (5S,7S,9S)- 9-[(2β,3β,4β,5α,12β,19α)- 3-(aminocarbonyl)- 3,4-dihydroxy- 16-methoxy- 1-methyl- 6,7-didehydroaspidospermidin- 15-yl]- 5-ethyl- 5-hydroxy- 1,4,5,6,7,8,9,10-octahydro- 2H- 3,7-methanoazacycloundecino[5,4-b]indole- 9-carboxylate
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEBI | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
| ECHA InfoCard | 100.053.330 |
|---|
|
| Formula | C43H55N5O7 |
|---|
| Molar mass | 753.941 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
O=C(OC)[C@]4(c2c(c1ccccc1[nH]2)CCN3C[C@](O)(CC)C[C@@H](C3)C4)c5c(OC)cc6c(c5)[C@@]89[C@@H](N6C)[C@@](O)(C(=O)N)[C@H](O)[C@@]7(/C=C\CN([C@@H]78)CC9)CC
|
InChI=1S/C43H55N5O7/c1-6-39(52)21-25-22-42(38(51)55-5,33-27(13-17-47(23-25)24-39)26-11-8-9-12-30(26)45-33)29-19-28-31(20-32(29)54-4)46(3)35-41(28)15-18-48-16-10-14-40(7-2,34(41)48)36(49)43(35,53)37(44)50/h8-12,14,19-20,25,34-36,45,49,52-53H,6-7,13,15-18,21-24H2,1-5H3,(H2,44,50)/t25-,34+,35-,36-,39+,40-,41-,42+,43+/m1/s1  Y YKey:HHJUWIANJFBDHT-KOTLKJBCSA-N  Y Y
|
| (verify) |
Vindesine, also termed Eldisine, is a semisynthetic vinca alkaloid derived from the flowering plant Catharanthus roseus.[1] Like the natural (e.g. vinblastine and vincristine) and semisynthetic vinca alkaloids (e.g. vinorelbine and vinflunine) derived from this plant, vindesine is an inhibitor of mitosis that is used as a chemotherapy drug.[2] By inhibiting mitosis, vinedsine blocks the proliferation of cells, particularly the rapidly proliferation cells of certain types of cancer. It is used, generally in combination with other chemotherapeutic drugs, in the treatment of various malignancies such as leukaemia, lymphoma, melanoma, breast cancer, and lung cancer.[3]
References
- ^ Mondal A, Gandhi A, Fimognari C, Atanasov AG, Bishayee A (September 2019). "Alkaloids for cancer prevention and therapy: Current progress and future perspectives". European Journal of Pharmacology. 858 172472. doi:10.1016/j.ejphar.2019.172472. PMID 31228447. S2CID 195298770.
- ^ Martino E, Casamassima G, Castiglione S, Cellupica E, Pantalone S, Papagni F, Rui M, Siciliano AM, Collina S (September 2018). "Vinca alkaloids and analogues as anti-cancer agents: Looking back, peering ahead". Bioorganic & Medicinal Chemistry Letters. 28 (17): 2816–2826. doi:10.1016/j.bmcl.2018.06.044. PMID 30122223. S2CID 52039135.
- ^ "Vindesine (Eldisine) | Cancer information | Cancer Research UK".
Intracellular chemotherapeutic agents / antineoplastic agents (L01) |
|---|
SPs/MIs
(M phase) | | Block microtubule assembly | |
|---|
| Block microtubule disassembly |
- Taxanes (Cabazitaxel
- Docetaxel#
- Larotaxel
- Ortataxel†
- Paclitaxel#
- Tesetaxel)
- Epothilones (Ixabepilone)
|
|---|
|
|---|
DNA replication
inhibitor | DNA precursors/
antimetabolites
(S phase) | | Folic acid | |
|---|
| Purine | |
|---|
| Pyrimidine |
- DNA polymerase inhibitor (Cytarabine# +daunorubicin)
|
|---|
| Deoxyribonucleotide | |
|---|
|
|---|
Topoisomerase inhibitors
(S phase) | | I |
- Camptotheca (Belotecan
- Camptothecin
- Cositecan†
- Etirinotecan pegol†
- Exatecan
- Gimatecan
- Irinotecan#
- Lurtotecan‡
- Rubitecan‡
- Silatecan§
- Topotecan)
|
|---|
| II | |
|---|
| II+Intercalation | |
|---|
|
|---|
Crosslinking of DNA
(CCNS) | | Alkylating | |
|---|
| Platinum-based | |
|---|
| Nonclassical | |
|---|
| Intercalation | |
|---|
|
|---|
|
|---|
| Photosensitizers/PDT | |
|---|
| Other | | Enzyme inhibitors |
- FI (Tipifarnib§)
- CDK inhibitors (Abemaciclib
- Alvocidib†
- Palbociclib
- Ribociclib
- Seliciclib†)
- PrI
- PhI (Anagrelide)
- IMPDI (Tiazofurin§)
- LI (Masoprocol)
- PARP inhibitor (Fuzuloparib
- Niraparib +abiraterone acetate
- Olaparib
- Rucaparib)
- HDAC (Belinostat
- Entinostat
- Panobinostat
- Romidepsin
- Vorinostat)
- PIKI (Pi3K) (Alpelisib
- Copanlisib‡
- Duvelisib
- Idelalisib
- Inavolisib
- Umbralisib‡)
- IDH (Enasidenib
- Ivosidenib
- Olutasidenib
- Vorasidenib)
|
|---|
| Receptor antagonists | |
|---|
| Other/ungrouped |
- Adagrasib
- Aflibercept
- Arsenic trioxide# (Realgar/Indigo naturalis#)
- Asparagine depleters (Asparaginase#/Pegaspargase)
- Axicabtagene ciloleucel
- Belzutifan
- Bexarotene
- Brexucabtagene autoleucel
- Calaspargase pegol
- Celecoxib
- Ciltacabtagene autoleucel
- Darinaparsin
- Demecolcine
- Denileukin diftitox
- Eflornithine
- Elesclomol§
- Elsamitrucin
- Epacadostat
- Eribulin
- Estramustine
- Glasdegib
- Idecabtagene vicleucel
- Idroxioleic acid
- Imetelstat
- Lifileucel
- Lisocabtagene maraleucel
- Lonidamine
- Lucanthone
- Lurbinectedin
- Mitoguazone
- Mitotane
- Nadofaragene firadenovec
- Navitoclax
- Nirogacestat
- Obecabtagene autoleucel
- Oblimersen†
- Omacetaxine mepesuccinate
- Para-toluenesulfonamide
- Pelabresib
- Plitidepsin
- Retinoids (Alitretinoin
- Tretinoin#)
- Selinexor
- Sitimagene ceradenovec
- Sotorasib
- Tabelecleucel
- Tagraxofusp
- Talimogene laherparepvec
- Tazemetostat
- Tebentafusp
- Tiazofurine
- Tigilanol tiglate
- Tisagenlecleucel
- Trabectedin
- Veliparib
- Venetoclax
- Verdinexor
- Vosaroxin
|
|---|
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|

