Valrubicin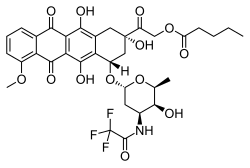 |
|
| AHFS/Drugs.com | Consumer Drug Information |
|---|
| MedlinePlus | a611021 |
|---|
Routes of
administration | Intravesical |
|---|
| ATC code | |
|---|
|
| Legal status |
- In general: ℞ (Prescription only)
|
|---|
|
| Bioavailability | Negligible |
|---|
| Protein binding | >99% |
|---|
| Metabolism | Negligible |
|---|
| Excretion | In urine |
|---|
|
2-oxo-2-[(2S,4S)-2,5,12-trihydroxy-7-methoxy-6,11-dioxo-4-({2,3,6-trideoxy-3-[(trifluoroacetyl)amino]hexopyranosyl}oxy)-1,2,3,4,6,11-hexahydrotetracen-2-yl]ethyl pentanoate
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
| ECHA InfoCard | 100.205.793 |
|---|
|
| Formula | C34H36F3NO13 |
|---|
| Molar mass | 723.651 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
FC(F)(F)C(=O)N[C@@H]5[C@H](O)[C@@H](O[C@@H](O[C@@H]4c3c(O)c2C(=O)c1c(OC)cccc1C(=O)c2c(O)c3C[C@@](O)(C(=O)COC(=O)CCCC)C4)C5)C
|
InChI=1S/C34H36F3NO13/c1-4-5-9-21(40)49-13-20(39)33(47)11-16-24(19(12-33)51-22-10-17(27(41)14(2)50-22)38-32(46)34(35,36)37)31(45)26-25(29(16)43)28(42)15-7-6-8-18(48-3)23(15)30(26)44/h6-8,14,17,19,22,27,41,43,45,47H,4-5,9-13H2,1-3H3,(H,38,46)/t14-,17-,19-,22-,27+,33-/m0/s1  Y YKey:ZOCKGBMQLCSHFP-KQRAQHLDSA-N  Y Y
|
| (verify) |
Valrubicin (N-trifluoroacetyladriamycin-14-valerate, trade name Valstar) is a chemotherapy drug used to treat bladder cancer.[1] Valrubicin is a semisynthetic analog of the anthracycline doxorubicin, and is administered by infusion directly into the bladder.
It was originally launched as Valstar in the U.S. in 1999 for intravesical therapy of Bacille Calmette-Guérin (BCG)-refractory carcinoma in situ of the urinary bladder in patients in whom cystectomy would be associated with unacceptable morbidity or mortality; however, it was voluntarily withdrawn in 2002 due to manufacturing issues.[2] Valstar was relaunched on September 3, 2009.[3]
Side effects
- Blood in urine
- Incontinence
- painful or difficult urination
- Unusually frequent urination
References
Intracellular chemotherapeutic agents / antineoplastic agents (L01) |
|---|
SPs/MIs
(M phase) | | Block microtubule assembly | |
|---|
| Block microtubule disassembly |
- Taxanes (Cabazitaxel
- Docetaxel#
- Larotaxel
- Ortataxel†
- Paclitaxel#
- Tesetaxel)
- Epothilones (Ixabepilone)
|
|---|
|
|---|
DNA replication
inhibitor | DNA precursors/
antimetabolites
(S phase) | | Folic acid | |
|---|
| Purine | |
|---|
| Pyrimidine |
- DNA polymerase inhibitor (Cytarabine# +daunorubicin)
|
|---|
| Deoxyribonucleotide | |
|---|
|
|---|
Topoisomerase inhibitors
(S phase) | | I |
- Camptotheca (Belotecan
- Camptothecin
- Cositecan†
- Etirinotecan pegol†
- Exatecan
- Gimatecan
- Irinotecan#
- Lurtotecan‡
- Rubitecan‡
- Silatecan§
- Topotecan)
|
|---|
| II | |
|---|
| II+Intercalation | |
|---|
|
|---|
Crosslinking of DNA
(CCNS) | | Alkylating | |
|---|
| Platinum-based | |
|---|
| Nonclassical | |
|---|
| Intercalation | |
|---|
|
|---|
|
|---|
| Photosensitizers/PDT | |
|---|
| Other | | Enzyme inhibitors |
- FI (Tipifarnib§)
- CDK inhibitors (Abemaciclib
- Alvocidib†
- Palbociclib
- Ribociclib
- Seliciclib†)
- PrI
- PhI (Anagrelide)
- IMPDI (Tiazofurin§)
- LI (Masoprocol)
- PARP inhibitor (Fuzuloparib
- Niraparib +abiraterone acetate
- Olaparib
- Rucaparib)
- HDAC (Belinostat
- Entinostat
- Panobinostat
- Romidepsin
- Vorinostat)
- PIKI (Pi3K) (Alpelisib
- Copanlisib‡
- Duvelisib
- Idelalisib
- Inavolisib
- Umbralisib‡)
- IDH (Enasidenib
- Ivosidenib
- Olutasidenib
- Vorasidenib)
|
|---|
| Receptor antagonists | |
|---|
| Other/ungrouped |
- Adagrasib
- Aflibercept
- Arsenic trioxide# (Realgar/Indigo naturalis#)
- Asparagine depleters (Asparaginase#/Pegaspargase)
- Axicabtagene ciloleucel
- Belzutifan
- Bexarotene
- Brexucabtagene autoleucel
- Calaspargase pegol
- Celecoxib
- Ciltacabtagene autoleucel
- Darinaparsin
- Demecolcine
- Denileukin diftitox
- Eflornithine
- Elesclomol§
- Elsamitrucin
- Epacadostat
- Eribulin
- Estramustine
- Glasdegib
- Idecabtagene vicleucel
- Idroxioleic acid
- Imetelstat
- Lifileucel
- Lisocabtagene maraleucel
- Lonidamine
- Lucanthone
- Lurbinectedin
- Mitoguazone
- Mitotane
- Nadofaragene firadenovec
- Navitoclax
- Nirogacestat
- Obecabtagene autoleucel
- Oblimersen†
- Omacetaxine mepesuccinate
- Para-toluenesulfonamide
- Pelabresib
- Plitidepsin
- Retinoids (Alitretinoin
- Tretinoin#)
- Selinexor
- Sitimagene ceradenovec
- Sotorasib
- Tabelecleucel
- Tagraxofusp
- Talimogene laherparepvec
- Tazemetostat
- Tebentafusp
- Tiazofurine
- Tigilanol tiglate
- Tisagenlecleucel
- Trabectedin
- Veliparib
- Venetoclax
- Verdinexor
- Vosaroxin
|
|---|
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
