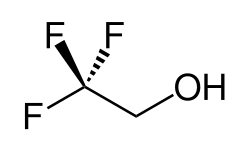
2,2,2-Trifluoroethanol
2,2,2-Trifluoroethanol
2,22-Trifluoroethanol
Names
Preferred IUPAC name
2,2,2-Trifluoroethan-1-ol
Other names
2,2,2-Trifluoroethanol
Identifiers
CAS Number
3D model (JSmol)
Beilstein Reference
1733203
ChEBI
ChEMBL
ChemSpider
DrugBank
ECHA InfoCard
100.000.831
EC Number
Gmelin Reference
2532
UNII
InChI=1S/C2H3F3O/c3-2(4,5)1-6/h6H,1H2
N Key: RHQDFWAXVIIEBN-UHFFFAOYSA-N
N InChI=1/C2H3F3O/c3-2(4,5)1-6/h6H,1H2
Key: RHQDFWAXVIIEBN-UHFFFAOYAH
Properties
Chemical formula
C2 H3 F3 O
Molar mass
100.04 g/mol
Appearance
Colorless liquid
Density
1.325±0.06 g/mL @ 20 °C, 760 Torr liquid
Melting point
−43.5 °C (−46.3 °F; 229.7 K)
Boiling point
74.0 °C (165.2 °F; 347.1 K)
Solubility in water
Miscible
Solubility in ethanol
Miscible
Acidity (pK a )
12.46±0.10 Most Acidic Temp: 25 °C
Viscosity
0.9 cSt @ 37.78 °C
Thermochemistry
Std enthalpy of(Δc H ⦵ 298 )
−886.6 kJ/mol
Hazards
GHS labelling:
Pictograms
Signal word
Danger
Hazard statements
H226 , H301 , H312 , H315 , H318 , H331 , H332 , H335 , H360 , H373
Precautionary statements
P201 , P202 , P210 , P233 , P240 , P241 , P242 , P243 , P260 , P264 , P270 , P271 , P280 , P281 , P301+P310 , P302+P352 , P303+P361+P353 , P304+P312 , P304+P340 , P305+P351+P338 , P308+P313 , P310 , P311 , P312 , P314 , P321 , P322 , P330 , P332+P313 , P362 , P363 , P370+P378 , P403+P233 , P403+P235 , P405 , P501
NFPA 704 (fire diamond)
Related compounds
Related alcohols
Hexafluoro-2-propanol
Related compounds
1,1,1-Trifluoroethane Trifluoroacetic acid
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
2,2,2-Trifluoroethanol is the synthetic organic compound with the formula CF3 CH2 OH. Also known as TFEA or trifluoroethyl alcohol , this colourless, water-miscible liquid has a smell reminiscent of ethanol . Due to the electronegativity of the trifluoromethyl group, this alcohol exhibits a stronger acidic character compared to ethanol.
Synthesis
Trifluoroethanol is produced industrially by hydrogenation or the hydride reduction of derivatives of trifluoroacetic acid , such as the esters or acyl chloride.[ 1]
TFEA can also be prepared by hydrogenolysis of compounds of generic formula CF3 −CHOH−OR (where R is hydrogen or an alkyl group containing from one to eight carbon atoms), in the presence of a palladium containing catalyst deposited on activated charcoal. As a co-catalyst for this conversion tertiary aliphatic amines like triethylamine are commonly employed.
Properties
Trifluoroethanol is used as a specialized solvent in organic chemistry.[ 2] [ 3] hydrogen peroxide are effectively conducted in TFEA.[ 4]
It competitively inhibits alcohol dehydrogenase for example.[ 5]
TFEA forms complexes with Lewis bases such as THF or pyridine through hydrogen bonding, yielding 1:1 adducts.[ 6] A = 2.07 and CA = 1.06.
TFEA can be used in biochemical experiments to stabilize alpha helix.[ 7] [ 8] [ 8]
Reactions
Oxidation of trifluoroethanol yields trifluoroacetic acid . It also serves as a source of the trifluoroethoxy group for various chemical reactions (Still-Gennari modification of HWE reaction).
2,2,2-Trifluoroethyl vinyl ether, an inhaled drug introduced clinically under the tradename Fluoromar, features a vinyl ether of trifluorethanol. This species was prepared by the reaction of trifluoroethanol with acetylene .[ 1]
Fluorester (2,2,2-trifluoroethyl methacrylate) is an ester of methacrylic acid and TFEA, which can be used as a monomer in specialty paints.[ 9]
Safety
Trifluoroethanol is classified as toxic to blood, the reproductive system, bladder, brain, upper respiratory tract and eyes.[ 10] [ 11]
See also
References
^ a b Siegemund G, Schwertfeger W, Feiring A, Smart B, Behr F, Vogel H, McKusick B, Kirsch P (2000). "Fluorine Compounds, Organic". Ullmann's Encyclopedia of Industrial Chemistry . John Wiley & Sons. doi:10.1002/14356007.a11_349 . ISBN 3527306730 . ^ Bégué JP, Bonnet-Delpon D, Crousse B (2004). "Fluorinated Alcohols: A New Medium for Selective and Clean Reaction". Synlett (Review) (1): 18– 29. doi:10.1055/s-2003-44973 . ^ Shuklov IA, Dubrovina NV, Börner A (2007). "Fluorinated Alcohols as Solvents, Cosolvents and Additives in Homogeneous Catalysis". Synthesis (Review). 2007 (19): 2925– 2943. doi:10.1055/s-2007-983902 . ^ Ravikumar KS, Kesavan V, Crousse B, Bonnet-Delpon D, Bégué JP (2003). "Mild and Selective Oxidation of Sulfur Compounds in Trifluorethanol: Diphenyl Disulfide and Methyl Phenyl Sulfoxide" . Organic Syntheses . 80 : 184. doi:10.15227/orgsyn.080.0184 ^ Taber RL (1998). "The competitive inhibition of yeast alcohol dehydrogenase by 2,2,2-trifluoroethanol". Biochemical Education . 26 (3): 239– 242. doi:10.1016/s0307-4412(98)00073-9 . ^ Sherry AD, Purcell KF (1970). "Linear enthalpy-spectral shift correlations for 2,2,2-trifluoroethanol". Journal of Physical Chemistry . 74 (19): 3535– 3543. Bibcode:1970JPhCh..74.3535S . doi:10.1021/j100713a017 . ^ Pereira AF, Piccoli V, Martínez L (2022-11-01). "Trifluoroethanol direct interactions with protein backbones destabilize α-helices" Journal of Molecular Liquids . 365 120209. doi:10.1016/j.molliq.2022.120209 . ISSN 0167-7322 . S2CID 251914912 . ^ a b Zhong L, Johnson WC (May 1992). "Environment affects amino acid preference for secondary structure" . Proceedings of the National Academy of Sciences of the United States of America . 89 (10): 4462– 4465. Bibcode:1992PNAS...89.4462Z . doi:10.1073/pnas.89.10.4462 49102 1584778 . ^ "New Flourine-Containing Monomer" . Paint & Coatings . BNP Media. September 26, 2000. Retrieved 20 November 2025 .^ "Sciencelab MSDS" . Archived from the original on 2016-03-03. Retrieved 2011-11-08 .^ Fischer Scientific MSDS
External links
Halocarbon Fluorochemicals Archived 2016-05-28 at the Wayback MachineUnited States Patent number 4,647,706 "Process for the synthesis of 2,2,2-Trifluoroethanol and 1,1,1,3,3,3-Hexafluoroisopropanol"
Hypnotics/sedatives (N05C)
GABAA
Alcohols Barbiturates Benzodiazepines Carbamates Imidazoles Monoureides
Acecarbromal
Apronal (apronalide)
Bromisoval
Capuride
Carbromal Ectylurea Neurosteroids Nonbenzodiazepines Phenols
Cipepofol (ciprofol)
Fospropofol
Propofol Propofol hemisuccinate Piperidinediones Quinazolinones
Afloqualone
Cloroqualone
Diproqualone
Etaqualone
Mebroqualone
Mecloqualone
Methaqualone Methylmethaqualone
Nitromethaqualone
SL-164 Others
GABAB H1
Antihistamines Antidepressants
Serotonin antagonists and reuptake inhibitors
Tricyclic antidepressants
Tetracyclic antidepressants
Antipsychotics
Typical antipsychotics
Atypical antipsychotics
α2 -Adrenergic 5-HT2A
Antidepressants
Trazodone Tricyclic antidepressants
Tetracyclic antidepressants
Antipsychotics
Typical antipsychotics
Atypical antipsychotics
Others
Melatonin Orexin
Daridorexant
Lemborexant
Suvorexant α2 δ VDCC Others
GABA A receptor positive modulators
Alcohols Barbiturates Benzodiazepines Carbamates
Carisbamate
Carisoprodol Clocental
Cyclarbamate
Difebarbamate
Emylcamate
Ethinamate Febarbamate
Felbamate Hexapropymate
Hydroxyphenamate
Lorbamate
Mebutamate
Meprobamate Nisobamate
Pentabamate
Phenprobamate
Procymate
Styramate
Tetrabamate
Tybamate Flavonoids Imidazoles
Etomidate Metomidate
Methoxyetomidate
Propoxate
Isopropoxate
Butomidate
Iso-butomidate
Sec-butomidate
CF2-Etomidate
CF3-Etomidate
CF3-Propoxate
Flutomidate
2,6-Dichloro-3-fluoroetomidate Kava constituents
10-Methoxyyangonin
11-Methoxyyangonin
11-Hydroxyyangonin
Desmethoxyyangonin
11-Methoxy-12-hydroxydehydrokavain
7,8-Dihydroyangonin
Kavain
5-Hydroxykavain
5,6-Dihydroyangonin
7,8-Dihydrokavain
5,6,7,8-Tetrahydroyangonin
5,6-Dehydromethysticin
Methysticin 7,8-Dihydromethysticin
Yangonin Monoureides
Acecarbromal
Apronal (apronalide)
Bromisoval
Carbromal Capuride
Ectylurea Neuroactive steroids Nonbenzodiazepines
Imidazopyridines : AlpidemDS-1
Necopidem
Saripidem
Zolpidem Pyrazolopyrimidines : DivaplonFasiplon
Indiplon
Lorediplon
Ocinaplon
Panadiplon
Taniplon
Zaleplon Others : AdipiplonAXS-17 (BAER-101, AZD-7325)
CGS-8216
CGS-9896
CGS-13767
CGS-20625
CL-218,872
CP-615,003
CTP-354
ELB-139
GBLD-345
Imepitoin
JM-1232
L-838,417
Lirequinil (Ro41-3696)
Miltirone (rosmariquinone)
NS-2664
NS-2710
NS-11394
Pipequaline
ROD-188
RWJ-51204
SB-205,384
SX-3228
TGSC01AA
TP-003
TPA-023
TP-13
U-89843A
U-90042
Viqualine
Y-23684 Phenols Piperidinediones Pyrazolopyridines
Cartazolate
Etazolate
ICI-190,622
Tracazolate Quinazolinones
Afloqualone
Cloroqualone
Diproqualone
Etaqualone
Mebroqualone
Mecloqualone
Methaqualone Methylmethaqualone
Nitromethaqualone
SL-164 Volatiles/gases Others/unsorted
3-Hydroxybutanal α-EMTBL
AA-29504
Alogabat
Avermectins (e.g., ivermectin )Bromide compounds (e.g., lithium bromide , potassium bromide , sodium bromide )
Carbamazepine Chloralose Chlormezanone Clomethiazole
Darigabat
DEABL
Deuterated etifoxine
Dihydroergolines (e.g., dihydroergocryptine, dihydroergosine, dihydroergotamine , ergoloid (dihydroergotoxine) )
DS2
Efavirenz Etazepine
Etifoxine
Fenamates (e.g., flufenamic acid , mefenamic acid , niflumic acid , tolfenamic acid )Fluoxetine Flupirtine
Hopantenic acid
KRM-II-81
Lanthanum Lavender oil
Lignans (e.g., 4-O-methylhonokiol, honokiol , magnolol , obovatol)
Loreclezole
Menthyl isovalerate (validolum) Monastrol Nicotinic acid Nicotinamide Org 25,435
Phenytoin Propanidid
Retigabine (ezogabine)
Safranal Seproxetine
Stiripentol
Sulfonylalkanes (e.g., sulfonmethane (sulfonal), tetronal, trional)
Terpenoids (e.g., borneol )
Topiramate Valerian constituents (e.g., isovaleric acid , isovaleramide, valerenic acid, valerenol) Unsorted benzodiazepine site positive modulators: α-Pinene MRK-409 (MK-0343)
TCS-1105
TCS-1205 See also: Receptor/signaling modulators • GABA receptor modulators • GABA metabolism/transport modulators
Glycine receptor modulators
Receptor(ligands)
GlyR Tooltip Glycine receptor
Positive modulators: Alcohols (e.g., brometone, chlorobutanol (chloretone) , ethanol (alcohol), tert -butanol (2M2P)tribromoethanol , trichloroethanol , trifluoroethanol )Alkylbenzene sulfonate Anandamide Barbiturates (e.g., pentobarbital , sodium thiopental )Chlormethiazole
D12-116
Dihydropyridines (e.g., nicardipine )
Etomidate Ginseng constituents (e.g., ginsenosides (e.g., ginsenoside-Rf))
Glutamic acid (glutamate) Ivermectin Ketamine Neuroactive steroids (e.g., alfaxolone, pregnenolone (eltanolone) , pregnenolone acetate , minaxolone, ORG-20599)
Nitrous oxide Penicillin G Propofol Tamoxifen Tetrahydrocannabinol Triclofos
Tropeines (e.g., atropine , bemesetron, cocaine , LY-278584, tropisetron, zatosetron)
Volatiles/gases (e.g., chloral hydrate , chloroform , desflurane , diethyl ether (ether) , enflurane , halothane , isoflurane , methoxyflurane , sevoflurane , toluene , trichloroethane (methyl chloroform) , trichloroethylene )
Xenon Zinc Antagonists: 2-Aminostrychnine2-Nitrostrychnine
4-Phenyl-4-formyl-N-methylpiperidine
αEMBTL
Bicuculline Brucine Cacotheline Caffeine Colchicine Colubrine
Cyanotriphenylborate
Dendrobine
Diaboline
Endocannabinoids (e.g., 2-AG, anandamide (AEA) )
Gaboxadol (THIP) Gelsemine iso-THAZ
Isobutyric acid Isonipecotic acid Isostrychnine
Laudanosine N-Methylbicuculline
N-Methylstrychnine
N,N-Dimethylmuscimol
Nipecotic acid Pitrazepin
Pseudostrychnine
Quinolines (e.g., 4-hydroxyquinoline , 4-hydroxyquinoline-3-carboxylic acid, 5,7-CIQA, 7-CIQ, 7-TFQ, 7-TFQA)RU-5135
Sinomenine Strychnine THAZ
Thiocolchicoside
Tutin Negative modulators: Amiloride Benzodiazepines (e.g., bromazepam , clonazepam , diazepam , flunitrazepam , flurazepam )
Corymine
Cyanotriphenylborate
Daidzein Dihydropyridines (e.g., nicardipine , nifedipine , nitrendipine )
Furosemide Genistein Ginkgo constituents (e.g., bilobalide, ginkgolides (e.g., ginkgolide A, ginkgolide B, ginkgolide C, ginkgolide J, ginkgolide M))
Imipramine NBQX
Neuroactive steroids (e.g., 3α-androsterone sulfate, 3β-androsterone sulfate, deoxycorticosterone , DHEA sulfate , pregnenolone sulfate, progesterone )
Opioids (e.g., codeine , dextromethorphan , dextrorphan, levomethadone, levorphanol , morphine , oripavine , pethidine , thebaine )
Picrotoxin (i.e., picrotin and picrotoxinin )PMBA
Riluzole Tropeines (e.g., bemesetron, LY-278584, tropisetron, zatosetron)
Verapamil Zinc NMDAR Tooltip N-Methyl-D-aspartate receptor
Transporter(blockers)
GlyT1 Tooltip Glycine transporter 1
ACPPB
ALX-5407 (NFPS)
ASP2535
Bitopertin (RG1678/RO4917838)
CP-802079
Ethanol (alcohol)
Glycyldodecylamide
GSK1018921
Iclepertin
LY-2365109
Mardepodect
ORG-24598
ORG-25935 (SCH-900435)
Pesampator (BIIB-104, PF-04958242)
PF-03463275
Sarcosine SNG-12 (Synapsinae)
SSR-103,800
SSR-504,734
Tilapertin GlyT2 Tooltip Glycine transporter 2
ALX-1393
Amoxapine Ethanol (alcohol)
NAGly
Opiranserin (VVZ-149)
ORG-25543
VVZ-368
See also
Receptor/signaling modulators GABA receptor modulators GABAA receptor positive modulators Ionotropic glutamate receptor modulators