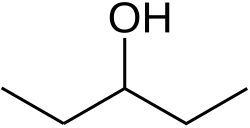
3-Pentanol[1]
|
|
| Names
|
| Preferred IUPAC name
|
| Other names
3-Pentanol, diethyl carbinol
|
| Identifiers
|
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI
|
|
| ChEMBL
|
|
| ChemSpider
|
|
| ECHA InfoCard
|
100.008.662
|
|
|
|
| UNII
|
|
|
|
|
InChI=1S/C5H12O/c1-3-5(6)4-2/h5-6H,3-4H2,1-2H3  Y YKey: AQIXEPGDORPWBJ-UHFFFAOYSA-N  Y YInChI=1/C5H12O/c1-3-5(6)4-2/h5-6H,3-4H2,1-2H3 Key: AQIXEPGDORPWBJ-UHFFFAOYAU
|
|
|
| Properties
|
Chemical formula
|
C5H12O
|
| Molar mass
|
88.148 g/mol
|
| Appearance
|
colorless liquid
|
| Density
|
0.815 g/ml
|
| Melting point
|
−63.68 °C (−82.62 °F; 209.47 K)
|
| Boiling point
|
115.3 °C (239.5 °F; 388.4 K)
|
Solubility in water
|
59 g/L
|
| Solubility
|
soluble in acetone, benzene; very soluble in ethanol, diethyl ether
|
| Vapor pressure
|
1.10 kPa
|
| Thermochemistry
|
Heat capacity (C)
|
2.719 J·g−1·K−1
|
Std enthalpy of
formation (ΔfH⦵298)
|
−368.9 kJ·mol−1 (liquid)
−314.9 kJ·mol−1 (gas)
|
| Hazards
|
| Flash point
|
41 °C (106 °F; 314 K)
|
Autoignition
temperature
|
435 °C (815 °F; 708 K)
|
| Explosive limits
|
1.2–9%
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
|
3-Pentanol is a secondary alcohol and one of the eight isomers of amyl alcohol. It is found naturally and has a role as an insect pheromone.[2]
See also
References
- ^
Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, Florida: CRC Press, pp. 3–454, 5–42, 8–102, 15–23, ISBN 0-8493-0594-2
- ^ PubChem. "3-Pentanol". pubchem.ncbi.nlm.nih.gov. Retrieved 2022-03-25.
Alcohols |
|---|
| By consumption | Alcohols found in
alcoholic drinks | |
|---|
| Medical alcohol | |
|---|
| Toxic alcohols | |
|---|
|
|---|
Primary
alcohols (1°) | | Methanol |
- 4-Methylcyclohexanemethanol
- Aminomethanol
- Cyclohexylmethanol
- Methoxymethanol
- Methylazoxymethanol
- Trifluoromethanol
|
|---|
| Ethanol | |
|---|
| Butanol | |
|---|
Straight-chain
saturated
C1 — C9 | |
|---|
Straight-chain
saturated
C10 — C19 | |
|---|
Straight-chain
saturated
C20 — C29 | |
|---|
Straight-chain
saturated
C30 — C39 |
- 1-Triacontanol (melissyl / myricyl)
- 1-Hentriacontanol
- 1-Dotriacontanol (lacceryl)
- 1-Tritriacontanol
- 1-Tetratriacontanol (geddyl)
- 1-Pentatriacontanol
- 1-Hexatriacontanol
- 1-Heptatriacontanol
- 1-Octatriacontanol
- 1-Nonatriacontanol
|
|---|
Straight-chain
saturated
C40 — C49 |
- 1-Tetracontanol
- 1-Hentetracontanol
- 1-Dotetracontanol
- 1-Tritetracontanol
- 1-Tetratetracontanol
- 1-Pentatetracontanol
- 1-Hexatetracontanol
- 1-Heptatetracontanol
- 1-Octatetracontanol
- 1-Nonatetracontanol
|
|---|
|
|---|
Secondary
alcohols (2°) | |
|---|
Tertiary
alcohols (3°) | |
|---|
| Hydric alcohols | |
|---|
| Amyl alcohols | |
|---|
| Aromatic alcohols | |
|---|
Saturated
fatty alcohols | |
|---|
Branched and
unsaturated
fatty alcohols | |
|---|
| Sugar alcohols | | C1 — C7 | |
|---|
Deoxy sugar
alcohols | |
|---|
Cyclic sugar
alcohols | |
|---|
| Glycylglycitols | |
|---|
|
|---|
| Terpene alcohols | Monoterpene
alcohols | |
|---|
Sesquiterpene
alcohols | |
|---|
Diterpene
alcohols | |
|---|
|
|---|
| Dialcohols | |
|---|
| Trialcohols | |
|---|
| Sterols | |
|---|
| Fluoroalcohols | |
|---|
| Preparations |
- Substitution of haloalkane
- Carbonyl reduction
- Ether cleavage
- Hydrolysis of epoxide
- Hydration of alkene
- Ziegler process
|
|---|
| Reactions |
- Deprotonation
- Protonation
- Alcohol oxidation
- Nucleophilic substitution
- Fischer–Speier esterification
- Williamson ether synthesis
- Elimination reaction
- Nucleophilic substitution of carbonyl group
- Friedel-Crafts alkylation
- Nucleophilic conjugate addition
- Transesterification
|
|---|
 Category Category
|

