Fucitol
Names
IUPAC name
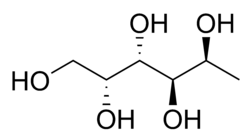
1-Deoxy-L -galactitol
Systematic IUPAC name
(2R ,3S ,4R ,5S )-Hexane-1,2,3,4,5-pentol
Identifiers
CAS Number
3D model (JSmol)
ChEBI
ChemSpider
UNII
InChI=1S/C6H14O5/c1-3(8)5(10)6(11)4(9)2-7/h3-11H,2H2,1H3/t3-,4+,5-,6-/m0/s1
N Key: SKCKOFZKJLZSFA-FSIIMWSLSA-N
N
C[C@H](O)[C@@H](O)[C@@H](O)[C@H](O)CO
Properties
Chemical formula
C 6 H 14 O 5
Molar mass
166.17 g/mol
Melting point
153 to 154 °C (307 to 309 °F; 426 to 427 K)[ 1]
Supplementary data page
Fucitol (data page)
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Fucitol , also known as L -fucitol1-deoxy-L -galactitol , and (2R ,3S ,4R ,5S )-hexane-1,2,3,4,5-pentol , is a sugar alcohol derived from fucoidan which is found in the North Atlantic seaweed Fucus vesiculosus [ 2] fucose .[ 1]
See also
References
External links
Fucitol at Wikimedia Commons
Alcohols
By consumption
Alcohols found in Medical alcohol Toxic alcohols
Primary
Methanol
4-Methylcyclohexanemethanol
Aminomethanol
Cyclohexylmethanol
Methoxymethanol
Methylazoxymethanol Trifluoromethanol Ethanol Butanol Straight-chain1 — C9 Straight-chain10 — C19 Straight-chain20 — C29 Straight-chain30 — C39
1-Triacontanol (melissyl / myricyl)1-Hentriacontanol
1-Dotriacontanol (lacceryl)1-Tritriacontanol
1-Tetratriacontanol (geddyl)
1-Pentatriacontanol
1-Hexatriacontanol
1-Heptatriacontanol
1-Octatriacontanol
1-Nonatriacontanol Straight-chain40 — C49
1-Tetracontanol
1-Hentetracontanol
1-Dotetracontanol
1-Tritetracontanol
1-Tetratetracontanol
1-Pentatetracontanol
1-Hexatetracontanol
1-Heptatetracontanol
1-Octatetracontanol
1-Nonatetracontanol
Secondary Tertiary Hydric alcohols
Amyl alcohols Aromatic alcohols Saturatedfatty alcohols Branched andfatty alcohols Sugar alcohols
C1 — C7 Deoxy sugar Cyclic sugar Glycylglycitols
Terpene alcohols
Monoterpene Sesquiterpene Diterpene
Dialcohols Trialcohols Sterols Fluoroalcohols Preparations
Substitution of haloalkane
Carbonyl reduction
Ether cleavage
Hydrolysis of epoxide
Hydration of alkene
Ziegler process Reactions
Deprotonation
Protonation Alcohol oxidation
Nucleophilic substitution
Fischer–Speier esterification
Williamson ether synthesis
Elimination reaction
Nucleophilic substitution of carbonyl group
Friedel-Crafts alkylation
Nucleophilic conjugate addition
Transesterification
 Media related to Fucitol at Wikimedia Commons
Media related to Fucitol at Wikimedia Commons