Gliquidone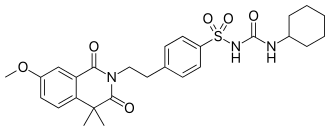 |
|
| Trade names | Glurenorm |
|---|
| AHFS/Drugs.com | International Drug Names |
|---|
Pregnancy
category | |
|---|
Routes of
administration | Oral (tablets) |
|---|
| ATC code | |
|---|
|
| Legal status |
- UK: POM (Prescription only)
- In general: ℞ (Prescription only)
|
|---|
|
| Bioavailability | High (Tmax = 2–3 hours) |
|---|
| Metabolism | Extensive hepatic |
|---|
| Onset of action | 1–1.5 hours |
|---|
| Excretion | Biliary (95%), renal (5%) |
|---|
|
1-cyclohexyl-3-[4-[2-(7-methoxy-4,4-dimethyl-1,3-dioxoisoquinolin-2-yl)ethyl]phenyl]sulfonylurea
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
| ECHA InfoCard | 100.046.770 |
|---|
|
| Formula | C27H33N3O6S |
|---|
| Molar mass | 527.64 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
O=C(NC1CCCCC1)NS(=O)(=O)c2ccc(cc2)CCN4C(=O)c3c(ccc(OC)c3)C(C4=O)(C)C
|
InChI=1S/C27H33N3O6S/c1-27(2)23-14-11-20(36-3)17-22(23)24(31)30(25(27)32)16-15-18-9-12-21(13-10-18)37(34,35)29-26(33)28-19-7-5-4-6-8-19/h9-14,17,19H,4-8,15-16H2,1-3H3,(H2,28,29,33)  Y YKey:LLJFMFZYVVLQKT-UHFFFAOYSA-N  Y Y
|
| (verify) |
Gliquidone (INN, sold under the trade name Glurenorm) is an anti-diabetic medication in the sulfonylurea class.[1] It is classified as a second-generation sulfonylurea. It is used in the treatment of diabetes mellitus type 2. It is marketed by the pharmaceutical company Boehringer Ingelheim (Germany).
Contraindications
- Allergy to sulfonylureas or sulfonamides
- Diabetes mellitus type 1
- Diabetic ketoacidosis
- Patients that underwent removal of the pancreas
- Acute porphyria
- Severe liver disease accompanying with liver insufficiency
- Several conditions (e.g., infectious diseases or major surgical intervention), when insulin administration is required
- Pregnancy or breastfeeding[2]
Pharmacokinetics
Gliquidone is fully metabolized by the liver. Its metabolites are excreted virtually completely with bile (even with long-term administration), thus allowing the use of medication in diabetic patients with kidney disease and diabetic nephropathy.[2]
References
Oral diabetes medication, insulins and insulin analogues, and other drugs used in diabetes (A10) |
|---|
|
|---|
| Fast-acting |
- Insulin aspart
- Insulin glulisine
- Insulin lispro
|
|---|
| Short-acting | |
|---|
| Long-acting |
- Insulin detemir#
- Insulin glargine# (+lixisenatide)
- NPH insulin
- Lente insulin‡
- Ultralente insulin‡
|
|---|
| Ultra-long-acting |
- Insulin degludec# (+insulin aspart, +liraglutide)
- Insulin efsitora†
- Insulin icodec† (+semaglutide)
|
|---|
| Inhalable | |
|---|
| Oral | |
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
|
Non-insulins |
|---|
| Insulin sensitizers | | Biguanides | |
|---|
| TZDs ("-glitazones") and PPAR agonists | |
|---|
| Dual PPAR agonists |
- Aleglitazar†
- Muraglitazar§
- Saroglitazar
- Tesaglitazar§
|
|---|
| Amylin analogues and DACRAs | |
|---|
|
|---|
| Secretagogues | | K+ATP | | Sulfonylureas | |
|---|
| Meglitinides ("-glinides") | |
|---|
|
|---|
| GLP-1 receptor agonists |
- Albiglutide‡
- Danuglipron†
- Dulaglutide
- Exenatide
- Liraglutide
- Lixisenatide
- Orforglipron†
- Semaglutide
- Taspoglutide†
| GLP1 poly-agonist peptides |
- Mazdutide (GLP-1/GCGR)
- Retatrutide§ (GLP-1/GIP/GCGR)
- Tirzepatide (GLP-1/GIP)
|
|---|
|
|---|
| DPP-4 inhibitors ("-gliptins") |
- Alogliptin
- Anagliptin
- Evogliptin
- Garvagliptin
- Gemigliptin
- Gosogliptin
- Linagliptin
- Melogliptin
- Omarigliptin
- Retagliptin
- Saxagliptin
- Sitagliptin
- Teneligliptin
- Trelagliptin
- Vildagliptin
|
|---|
|
|---|
| Other | | Aldose reductase inhibitors | |
|---|
| Alpha-glucosidase inhibitors | |
|---|
| SGLT2 inhibitors ("-gliflozins") |
- Canagliflozin
- Dapagliflozin
- Empagliflozin#
- Ertugliflozin
- Ipragliflozin
- Luseogliflozin
- Remogliflozin§
- Sergliflozin§
- Sotagliflozin
- Tofogliflozin†
- Velagliflozin
|
|---|
| Other | |
|---|
|
|---|
| Combinations |
- Alogliptin/metformin
- Canagliflozin/metformin
- Cagrilintide/semaglutide§
- Dapagliflozin/metformin
- Dapagliflozin/saxagliptin
- Dapagliflozin/saxagliptin/metformin
- Empagliflozin/linagliptin
- Empagliflozin/metformin
- Gemigliptin/rosuvastatin
- Glibenclamide (glyburide)/metformin
- Glimepiride/rosiglitazone
- Linagliptin/metformin
- Metformin/acarbose
- Metformin/ertugliflozin
- Metformin/evogliptin
- Metformin/gemigliptin
- Metformin/repaglinide
- Metformin/sulfonylureas
- Metformin/teneligliptin
- Phenformin/sulfonylureas
- Pioglitazone/alogliptin
- Pioglitazone/glimepiride
- Pioglitazone/metformin
- Pioglitazone/sitagliptin
- Rosiglitazone/metformin
- Saxagliptin/metformin
- Sitagliptin/dapagliflozin
- Sitagliptin/ertugliflozin
- Sitagliptin/metformin
- Sitagliptin/simvastatin
- Vildagliptin/metformin
|
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
|
