Glisoxepide
|
|
| Names
|
Preferred IUPAC name
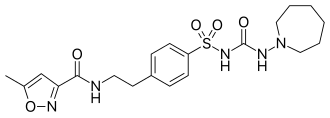
N-[2-(4-{[(Azepan-1-yl)carbamoyl]sulfamoyl}phenyl)ethyl]-5-methyl-1,2-oxazole-3-carboxamide |
| Identifiers
|
CAS Number
|
|
3D model (JSmol)
|
|
| ChEMBL
|
|
| ChemSpider
|
|
| DrugBank
|
|
| ECHA InfoCard
|
100.042.329
|
| KEGG
|
|
|
|
|
| UNII
|
|
|
|
|
InChI=1S/C20H27N5O5S/c1-15-14-18(23-30-15)19(26)21-11-10-16-6-8-17(9-7-16)31(28,29)24-20(27)22-25-12-4-2-3-5-13-25/h6-9,14H,2-5,10-13H2,1H3,(H,21,26)(H2,22,24,27)  Y YKey: ZKUDBRCEOBOWLF-UHFFFAOYSA-N  Y YInChI=1/C20H27N5O5S/c1-15-14-18(23-30-15)19(26)21-11-10-16-6-8-17(9-7-16)31(28,29)24-20(27)22-25-12-4-2-3-5-13-25/h6-9,14H,2-5,10-13H2,1H3,(H,21,26)(H2,22,24,27) Key: ZKUDBRCEOBOWLF-UHFFFAOYAG
|
O=C(NCCc1ccc(cc1)S(=O)(=O)NC(=O)NN2CCCCCC2)c3noc(c3)C
|
| Properties
|
Chemical formula
|
C20H27N5O5S
|
| Molar mass
|
449.52388 g/mol
|
| Pharmacology
|
ATC code
|
A10BB11 (WHO)
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
|
Glisoxepide (INN) is an orally available anti-diabetic drug from the group of sulfonylureas.[1] It belongs to second-generation sulfonylureas.[2]
References
Oral diabetes medication, insulins and insulin analogues, and other drugs used in diabetes (A10) |
|---|
|
|---|
| Fast-acting |
- Insulin aspart
- Insulin glulisine
- Insulin lispro
|
|---|
| Short-acting | |
|---|
| Long-acting |
- Insulin detemir#
- Insulin glargine# (+lixisenatide)
- NPH insulin
- Lente insulin‡
- Ultralente insulin‡
|
|---|
| Ultra-long-acting |
- Insulin degludec# (+insulin aspart, +liraglutide)
- Insulin efsitora†
- Insulin icodec† (+semaglutide)
|
|---|
| Inhalable | |
|---|
| Oral | |
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
|
Non-insulins |
|---|
| Insulin sensitizers | | Biguanides | |
|---|
| TZDs ("-glitazones") and PPAR agonists | |
|---|
| Dual PPAR agonists |
- Aleglitazar†
- Muraglitazar§
- Saroglitazar
- Tesaglitazar§
|
|---|
| Amylin analogues and DACRAs | |
|---|
|
|---|
| Secretagogues | | K+ATP | | Sulfonylureas | |
|---|
| Meglitinides ("-glinides") | |
|---|
|
|---|
| GLP-1 receptor agonists |
- Albiglutide‡
- Danuglipron†
- Dulaglutide
- Exenatide
- Liraglutide
- Lixisenatide
- Orforglipron†
- Semaglutide
- Taspoglutide†
| GLP1 poly-agonist peptides |
- Mazdutide (GLP-1/GCGR)
- Retatrutide§ (GLP-1/GIP/GCGR)
- Tirzepatide (GLP-1/GIP)
|
|---|
|
|---|
| DPP-4 inhibitors ("-gliptins") |
- Alogliptin
- Anagliptin
- Evogliptin
- Garvagliptin
- Gemigliptin
- Gosogliptin
- Linagliptin
- Melogliptin
- Omarigliptin
- Retagliptin
- Saxagliptin
- Sitagliptin
- Teneligliptin
- Trelagliptin
- Vildagliptin
|
|---|
|
|---|
| Other | | Aldose reductase inhibitors | |
|---|
| Alpha-glucosidase inhibitors | |
|---|
| SGLT2 inhibitors ("-gliflozins") |
- Canagliflozin
- Dapagliflozin
- Empagliflozin#
- Ertugliflozin
- Ipragliflozin
- Luseogliflozin
- Remogliflozin§
- Sergliflozin§
- Sotagliflozin
- Tofogliflozin†
- Velagliflozin
|
|---|
| Other | |
|---|
|
|---|
| Combinations |
- Alogliptin/metformin
- Canagliflozin/metformin
- Cagrilintide/semaglutide§
- Dapagliflozin/metformin
- Dapagliflozin/saxagliptin
- Dapagliflozin/saxagliptin/metformin
- Empagliflozin/linagliptin
- Empagliflozin/metformin
- Gemigliptin/rosuvastatin
- Glibenclamide (glyburide)/metformin
- Glimepiride/rosiglitazone
- Linagliptin/metformin
- Metformin/acarbose
- Metformin/ertugliflozin
- Metformin/evogliptin
- Metformin/gemigliptin
- Metformin/repaglinide
- Metformin/sulfonylureas
- Metformin/teneligliptin
- Phenformin/sulfonylureas
- Pioglitazone/alogliptin
- Pioglitazone/glimepiride
- Pioglitazone/metformin
- Pioglitazone/sitagliptin
- Rosiglitazone/metformin
- Saxagliptin/metformin
- Sitagliptin/dapagliflozin
- Sitagliptin/ertugliflozin
- Sitagliptin/metformin
- Sitagliptin/simvastatin
- Vildagliptin/metformin
|
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
|

