Mitiglinide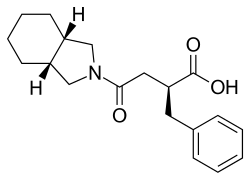 |
|
| Trade names | Glufast |
|---|
| AHFS/Drugs.com | International Drug Names |
|---|
Routes of
administration | By mouth (tablets) |
|---|
| ATC code | |
|---|
|
(−)-(2S,3a,7a-cis)-α-Benzylhexahydro-γ-oxo-2-isoindolinebutyric acid
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
|
| Formula | C19H25NO3 |
|---|
| Molar mass | 315.413 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
O=C(O)[C@@H](Cc1ccccc1)CC(=O)N3C[C@H]2CCCC[C@H]2C3
|
InChI=1S/C19H25NO3/c21-18(20-12-15-8-4-5-9-16(15)13-20)11-17(19(22)23)10-14-6-2-1-3-7-14/h1-3,6-7,15-17H,4-5,8-13H2,(H,22,23)/t15-,16+,17-/m0/s1  Y YKey:WPGGHFDDFPHPOB-BBWFWOEESA-N  Y Y
|
 N N Y (what is this?) (verify) Y (what is this?) (verify) |
Mitiglinide (INN,[1] trade name Glufast) is a drug for the treatment of type 2 diabetes.[2]
Mitiglinide belongs to the meglitinide (glinide) class of blood glucose-lowering drugs and is currently co-marketed in Japan by Kissei and Takeda. The North America rights to mitiglinide are held by Elixir Pharmaceuticals. Mitiglinide has not yet gained FDA approval.
Pharmacology
Mitiglinide is thought to stimulate insulin secretion by closing the ATP-sensitive potassium KATP channels in pancreatic β cells.
Dosage
Mitiglinide is delivered in tablet form.
References
External links
Oral diabetes medication, insulins and insulin analogues, and other drugs used in diabetes (A10) |
|---|
|
|---|
| Fast-acting |
- Insulin aspart
- Insulin glulisine
- Insulin lispro
|
|---|
| Short-acting | |
|---|
| Long-acting |
- Insulin detemir#
- Insulin glargine# (+lixisenatide)
- NPH insulin
- Lente insulin‡
- Ultralente insulin‡
|
|---|
| Ultra-long-acting |
- Insulin degludec# (+insulin aspart, +liraglutide)
- Insulin efsitora†
- Insulin icodec† (+semaglutide)
|
|---|
| Inhalable | |
|---|
| Oral | |
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
|
Non-insulins |
|---|
| Insulin sensitizers | | Biguanides | |
|---|
| TZDs ("-glitazones") and PPAR agonists | |
|---|
| Dual PPAR agonists |
- Aleglitazar†
- Muraglitazar§
- Saroglitazar
- Tesaglitazar§
|
|---|
| Amylin analogues and DACRAs | |
|---|
|
|---|
| Secretagogues | | K+ATP | | Sulfonylureas | |
|---|
| Meglitinides ("-glinides") | |
|---|
|
|---|
| GLP-1 receptor agonists |
- Albiglutide‡
- Danuglipron†
- Dulaglutide
- Exenatide
- Liraglutide
- Lixisenatide
- Orforglipron†
- Semaglutide
- Taspoglutide†
| GLP1 poly-agonist peptides |
- Mazdutide (GLP-1/GCGR)
- Retatrutide§ (GLP-1/GIP/GCGR)
- Tirzepatide (GLP-1/GIP)
|
|---|
|
|---|
| DPP-4 inhibitors ("-gliptins") |
- Alogliptin
- Anagliptin
- Evogliptin
- Garvagliptin
- Gemigliptin
- Gosogliptin
- Linagliptin
- Melogliptin
- Omarigliptin
- Retagliptin
- Saxagliptin
- Sitagliptin
- Teneligliptin
- Trelagliptin
- Vildagliptin
|
|---|
|
|---|
| Other | | Aldose reductase inhibitors | |
|---|
| Alpha-glucosidase inhibitors | |
|---|
| SGLT2 inhibitors ("-gliflozins") |
- Canagliflozin
- Dapagliflozin
- Empagliflozin#
- Ertugliflozin
- Ipragliflozin
- Luseogliflozin
- Remogliflozin§
- Sergliflozin§
- Sotagliflozin
- Tofogliflozin†
- Velagliflozin
|
|---|
| Other | |
|---|
|
|---|
| Combinations |
- Alogliptin/metformin
- Canagliflozin/metformin
- Cagrilintide/semaglutide§
- Dapagliflozin/metformin
- Dapagliflozin/saxagliptin
- Dapagliflozin/saxagliptin/metformin
- Empagliflozin/linagliptin
- Empagliflozin/metformin
- Gemigliptin/rosuvastatin
- Glibenclamide (glyburide)/metformin
- Glimepiride/rosiglitazone
- Linagliptin/metformin
- Metformin/acarbose
- Metformin/ertugliflozin
- Metformin/evogliptin
- Metformin/gemigliptin
- Metformin/repaglinide
- Metformin/sulfonylureas
- Metformin/teneligliptin
- Phenformin/sulfonylureas
- Pioglitazone/alogliptin
- Pioglitazone/glimepiride
- Pioglitazone/metformin
- Pioglitazone/sitagliptin
- Rosiglitazone/metformin
- Saxagliptin/metformin
- Sitagliptin/dapagliflozin
- Sitagliptin/ertugliflozin
- Sitagliptin/metformin
- Sitagliptin/simvastatin
- Vildagliptin/metformin
|
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
|
