Tolazamide Trade names Tolinase AHFS/Drugs.com Monograph MedlinePlus a682482 License data
Pregnancy Routes of Oral ATC code Legal status
Bioavailability ? Metabolism metabolized in the liver to active metabolites Elimination half-life 7 hours Excretion Renal (85%) and fecal (7%)
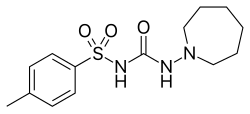
N -[(azepan-1-ylamino)carbonyl]-4-methylbenzenesulfonamide
CAS Number PubChem CID IUPHAR/BPS DrugBank ChemSpider UNII KEGG ChEBI ChEMBL CompTox Dashboard (EPA ) ECHA InfoCard 100.013.262 Formula C 14 H 21 N 3 O 3 S Molar mass −1 3D model (JSmol)
O=S(=O)(c1ccc(cc1)C)NC(=O)NN2CCCCCC2
InChI=1S/C14H21N3O3S/c1-12-6-8-13(9-7-12)21(19,20)16-14(18)15-17-10-4-2-3-5-11-17/h6-9H,2-5,10-11H2,1H3,(H2,15,16,18)
Y Key:OUDSBRTVNLOZBN-UHFFFAOYSA-N
Y (verify)
Tolazamide is an oral blood glucose lowering drug used for people with Type 2 diabetes. It is part of the sulfonylurea family (ATC A10BB).
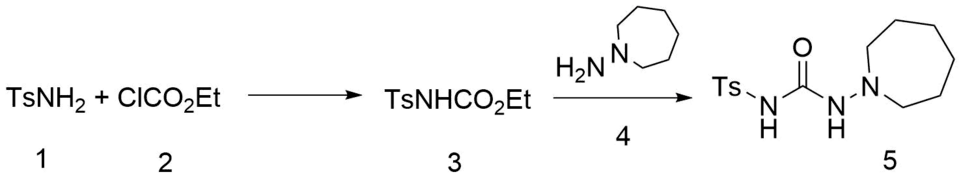
Synthesis
The reaction between p-toluenesulfonamide (1 ) and ethyl chloroformate (2 ) in the presence of base gives tosylurethane [5577-13-9] (3 ). Heating that intermediate with azepane (4 ) leads to the displacement of the ethoxy group and the formation of tolazemide (5 ).[ 1] [ 2] [ 3] [ 4] [ 5]
Azepane proper would lead to [13078-23-4].
References
^ Vardanyan, Ṛuben, Hruby, V. J. (2006). Synthesis of essential drugs . Elsevier. ISBN 9780444521668 . ^ Wright JB, Willette RE (July 1962). "Antidiabetic Agents. N4-Arylsulfonylsemicarbazides". Journal of Medicinal and Pharmaceutical Chemistry . 91 (4): 815– 22. doi:10.1021/jm01239a016 . PMID 14056414 . ^ John B Wright, U.S. patent 3,063,903
^ Wright John Brenton, GB 887886
^ DE1196200 idem Korger Gerhard, Weber Helmut, Aumuller Walter, U.S. patent 3,248,384
External links
"Tolazamide" . Medline Plus . U.S. National Library of Medicine.
Oral diabetes medication,
insulins and insulin analogues, and other drugs used in diabetes (A10)
Fast-acting
Insulin aspart
Insulin glulisine
Insulin lispro Short-acting Long-acting
Insulin detemir#
Insulin glargine# (+lixisenatide)
NPH insulin
Lente insulin‡
Ultralente insulin‡ Ultra-long-acting
Insulin degludec# (+insulin aspart, +liraglutide)
Insulin efsitora†
Insulin icodec† (+semaglutide) Inhalable Oral
# WHO-EM‡ Withdrawn from marketClinical trials:
† Phase III§ Never to phase III
Non-insulins
Insulin sensitizers
Biguanides TZDs ("-glitazones") and PPAR agonistsDual PPAR agonists
Aleglitazar†
Muraglitazar§
Saroglitazar
Tesaglitazar§ Amylin analogues and DACRAs
Secretagogues
K+ ATP
Sulfonylureas Meglitinides ("-glinides")
GLP-1 receptor agonists
Albiglutide‡
Danuglipron†
Dulaglutide
Exenatide Liraglutide
Lixisenatide
Orforglipron†
Semaglutide
Taspoglutide† GLP1 poly-agonist peptides
Mazdutide (GLP-1/GCGR)
Retatrutide§ (GLP-1/GIP/GCGR)
Tirzepatide (GLP-1/GIP)
DPP-4 inhibitors ("-gliptins")
Alogliptin
Anagliptin
Evogliptin
Garvagliptin
Gemigliptin
Gosogliptin
Linagliptin
Melogliptin
Omarigliptin
Retagliptin
Saxagliptin
Sitagliptin Teneligliptin
Trelagliptin
Vildagliptin
Other
Aldose reductase inhibitors Alpha-glucosidase inhibitors SGLT2 inhibitors ("-gliflozins")
Canagliflozin
Dapagliflozin
Empagliflozin#
Ertugliflozin
Ipragliflozin
Luseogliflozin
Remogliflozin§
Sergliflozin§
Sotagliflozin
Tofogliflozin†
Velagliflozin Other
Combinations
Alogliptin/metformin
Canagliflozin/metformin
Cagrilintide/semaglutide§
Dapagliflozin/metformin
Dapagliflozin/saxagliptin
Dapagliflozin/saxagliptin/metformin
Empagliflozin/linagliptin
Empagliflozin/metformin
Gemigliptin/rosuvastatin
Glibenclamide (glyburide)/metformin
Glimepiride/rosiglitazone
Linagliptin/metformin Metformin/acarbose
Metformin/ertugliflozin
Metformin/evogliptin
Metformin/gemigliptin
Metformin/repaglinide
Metformin/sulfonylureas
Metformin/teneligliptin
Phenformin/sulfonylureas
Pioglitazone/alogliptin
Pioglitazone/glimepiride
Pioglitazone/metformin
Pioglitazone/sitagliptin
Rosiglitazone/metformin Saxagliptin/metformin Sitagliptin/dapagliflozin
Sitagliptin/ertugliflozin
Sitagliptin/metformin
Sitagliptin/simvastatin
Vildagliptin/metformin
# WHO-EM‡ Withdrawn from marketClinical trials:
† Phase III§ Never to phase III