n-Propylbenzene
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Propylbenzene | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.848 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 2364 (N-PROPYL BENZENE) |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C9H12 |
| Molar mass | 120.195 g·mol−1 |
| Appearance | colorless liquid |
| Density | 0.8620 g/cm3 |
| Melting point | −99.5 °C (−147.1 °F; 173.7 K) |
| Boiling point | 159.2 °C (318.6 °F; 432.3 K) |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|
   
|
Signal word
|
Danger |
Hazard statements
|
H226, H304, H335, H411 |
Precautionary statements
|
P210, P233, P240, P241, P242, P243, P261, P271, P273, P280, P301+P316, P303+P361+P353, P304+P340, P319, P331, P370+P378, P391, P403+P233, P403+P235, P405, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
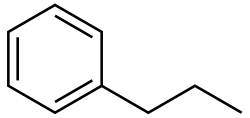
n-Propylbenzene is an aromatic hydrocarbon with the formula C
6H
5CH
2CH
2CH
3. The molecule consists of a propyl group attached to a phenyl ring. It is a colorless liquid. A more common structural isomer of this compound is cumene.
n-Propylbenzene is used as a nonpolar organic solvent in various industries, including printing and the dyeing of textiles and in the manufacture of methylstyrene.[1][2] It can be synthesized by the reaction of the Grignard reagent derived from benzyl chloride with diethyl sulfate.[3]
References
- ^ Cleland, J. G. (1979). Multimedia Environmental Goals for Environmental Assessment: MEG charts and background information summaries (categories 13–26). Environmental Protection Agency, Office of Research and Development, Office of Energy, Minerals and Industry, Industrial Environmental Research Laboratory. pp. A528–A529.
- ^ Montgomery, John H. (1991). Groundwater Chemicals Field Guide. CRC Press. p. 174.
- ^ Henry Gilman and W. E. Catlin (1941). "n-Propylbenzene". Organic Syntheses; Collected Volumes, vol. 1, p. 471.