Osmium pentafluoride
 | |
 | |
| Identifiers | |
|---|---|
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
F5Os |
| Molar mass | 285.22 g·mol−1 |
| Appearance | blue-green solid |
| Melting point | 70 °C (158 °F; 343 K) |
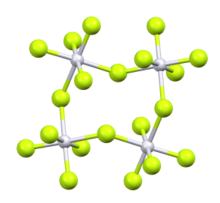
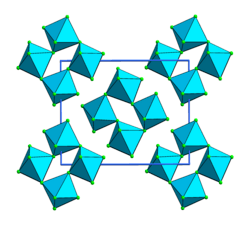
| Structure[1] | |
Crystal structure
|
Monoclinic |
Space group
|
P21/c (No. 14) |
Lattice constant
|
a = 5.53 Å, b = 9.91 Å, c = 12.59 Å α = 90°, β = 99.5°, γ = 90°
|
Lattice volume (V)
|
680 Å3 |
Formula units (Z)
|
8 units per cell |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Osmium pentafluoride is an inorganic compound with the formula OsF5. It is a blue-green solid. Like the pentafluorides of Ru, Rh, and Ir, OsF5 exists as a tetramer in the solid state.
Preparation
Osmium pentafluoride can be prepared by reduction of osmium hexafluoride with iodine as a solution in iodine pentafluoride:[1]
- 10 OsF6 + I2 → 10 OsF5 + 2 IF5
It can also be prepared by treating a solution of osmium hexafluoride in anhydrous hydrogen fluoride with hydrogen or silicon.[2] Platinum gauze or UV irradiation increase the reaction rate.
References
- ^ a b Holloway, John H.; Mitchell, S. J. (1971). "Preparation and Crystal Structure of Osmium Pentafluoride". Journal of the Chemical Society: 2789–94. doi:10.1039/J19710002789.
- ^ Paine, Robert T.; Asprey, Larned B. (1975-05-01). "Reductive syntheses of transition metal fluoride compounds. Synthesis of rhenium, osmium, and iridium pentafluorides and tetrafluorides". Inorganic Chemistry. 14 (5): 1111–1113. doi:10.1021/ic50147a030. ISSN 0020-1669.