Perbromyl fluoride
 | |
| Names | |
|---|---|
| Other names
Bromine fluoride trioxide
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
PubChem CID
|
|
InChI
| |
SMILES
| |
| Properties | |
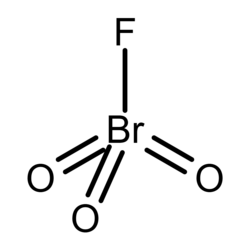
Chemical formula
|
BrO3F |
| Molar mass | 146.899 g·mol−1 |
| Appearance | colorless gas |
| Melting point | −110 °C (−166 °F; 163 K)[1] (approx. temperature of solidification) |
Solubility in water
|
reacts with water |
| Related compounds | |
Related compounds
|
Periodyl fluoride Perchloryl fluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Perbromyl fluoride is an inorganic compound of bromine, fluorine, and oxygen with the chemical formula BrO3F.[1]
Synthesis
Synthesis of perbromyl fluoride is by the effect of antimony pentafluoride on a solution of potassium perbromate in hydrofluoric acid:[2]
- KBrO4 + 2 SbF5 + 3 HF → BrO3F + K[SbF6] + H[SbF6]·H2O
Physical properties
Perbromyl fluoride is a colorless gas at room temperature that is stable in the absence of moisture.[3]
Chemical properties
Perbromyl fluoride reacts with water to produce perbromic acid and hydrogen fluoride:
- BrO3F + H2O → HBrO4 + HF
References
- ^ a b Appleman, Evan H.; Studier, Martin H. (July 1969). "Perbromyl fluoride". Journal of the American Chemical Society. 91 (16): 4561–4562. Bibcode:1969JAChS..91.4561A. doi:10.1021/ja01044a049. ISSN 0002-7863.
- ^ Steudel, Ralf (20 April 2011). Chemistry of the Non-Metals: With an Introduction to Atomic Structure and Chemical Bonding. Walter de Gruyter. p. 265. ISBN 978-3-11-083082-8. Retrieved 5 June 2023.
- ^ U. S. Atomic Energy Commission Division of Plans and Atomic Energy Research: Life and Physical Sciences, Reactor Development, Waste Management. U.S. Government Printing Office. 1970. p. 10. Retrieved 5 June 2023.