Sulfuryl bromide fluoride
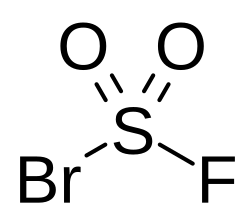 | |
| Names | |
|---|---|
| Preferred IUPAC name
Sulfuryl bromide fluoride | |
| Other names
Sulfuryl bromofluoride, sulfur bromide fluoride oxide
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
BrFO2S |
| Molar mass | 162.96 g·mol−1 |
| Appearance | Colorless liquid |
| Melting point | −86 °C (−123 °F; 187 K) |
| Boiling point | 41 °C (106 °F; 314 K) |
Solubility in water
|
Reacts with water |
| Hazards | |
| Flash point | −273.15 °C (−459.67 °F; 0.00 K) |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Sulfuryl bromide fluoride is an inorganic compound of bromine, fluorine, oxygen, and sulfur with the chemical formula BrFO2S.[1][2][3]
Synthesis
Synthesis of sulfuryl bromide fluoride can be by a reaction of bromine, bromine trifluoride, and sulfur dioxide:[4][5]
- Br2 + BrF3 → 3 BrF
- SO2 + BrF → SO2BrF
Physical properties
Sulfuryl fluoride bromide forms a colorless liquid that, when standing in humid air, turns slightly red due to the release of elemental bromine.[6]
Sulfuryl bromide fluoride reacts slowly with glass, but has no effect on quartz.[5]
Chemical properties
Sulfuryl bromide fluoride violently reacts with water:[5]
- SO2BrF + 2 H2O → H2SO4 + HBr + HF
References
- ^ "Sulfuryl bromide fluoride". NIST. Retrieved 12 January 2026.
- ^ Raley, John M.; Wollrab, James E.; Lovejoy, R. W. (1 October 1973). "Rotational spectrum of sulfuryl bromide fluoride". Journal of Molecular Spectroscopy. 48 (1): 100–106. Bibcode:1973JMoSp..48..100R. doi:10.1016/0022-2852(73)90138-0. ISSN 0022-2852. Retrieved 12 January 2026.
- ^ Kuchitsu, Kozo (9 March 2013). Structure of Free Polyatomic Molecules: Basic Data. Springer Science & Business Media. p. 31. ISBN 978-3-642-45748-7. Retrieved 12 January 2026.
- ^ "Sulfuryl bromide fluoride | 13536-61-3". ChemicalBook. Retrieved 12 January 2026.
- ^ a b c Brauer, Georg (2 December 2012). Handbook of Preparative Inorganic Chemistry V1. Elsevier. p. 176-177. ISBN 978-0-323-16127-5. Retrieved 12 January 2026.
- ^ Haynes, William M. (19 April 2016). CRC Handbook of Chemistry and Physics, 94th Edition. CRC Press. p. 4-93. ISBN 978-1-4665-7115-0. Retrieved 12 January 2026.