Fluoroboric acid
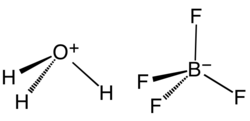 Hydronium tetrafluoroborate
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Tetrafluoroboric acid[1] | |||
Other names
| |||
| Identifiers | |||
CAS Number
|
| ||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.037.165 | ||
| EC Number |
| ||
Gmelin Reference
|
21702 | ||
| MeSH | Fluoroboric+acid | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1775 | ||
CompTox Dashboard (EPA)
|
|||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula
|
H[BF4] | ||
| Molar mass | 87.81 g·mol−1 | ||
| Appearance | Colourless liquid | ||
| Density | 1.4 g/mL (48% solution in water)[2] | ||
| Melting point | −90 °C (−130 °F; 183 K)[4] | ||
| Boiling point | 130 °C (266 °F; 403 K)[4] | ||
| Vapor pressure | 5 mmHg (0.67 kPa) (20 °C (68 °F; 293 K))[2] | ||
| Acidity (pKa) | ~1.8 (MeCN solution)[3] | ||
| Hazards | |||
| GHS labelling:[2] | |||
Pictograms
|

| ||
Signal word
|
Danger | ||
Hazard statements
|
H314 | ||
Precautionary statements
|
P264, P280, P301+P330+P331, P303+P361+P353, P304+P340+P310, P305+P351+P338+P310, P405, P501 | ||
| NFPA 704 (fire diamond) | 
3
1
0 | ||
Threshold limit value (TLV)
|
2.5 mg/m3 (TWA), 6 mg/m3[4] (STEL) | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
2.5 mg/m3 (TWA) | ||
IDLH (Immediate danger)
|
250 mg/m3 | ||
| Related compounds | |||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |||
Fluoroboric acid or tetrafluoroboric acid (archaically, fluoboric acid) is an inorganic compound with the simplified chemical formula H+[BF4]−. Solvent-free tetrafluoroboric acid (H[BF4]) has not been reported. The term "fluoroboric acid" usually refers to a range of compounds including hydronium tetrafluoroborate ([H3O]+[BF4]−), which are available as solutions. The ethyl ether solvate is also commercially available, where the fluoroboric acid can be represented by the formula [H((CH3CH2)2O)n]+[BF4]−, where n is 2.
It is mainly produced as a precursor to other fluoroborate salts.[5] It is a strong acid. Fluoroboric acid is corrosive and attacks the skin. It is available commercially as a solution in water and other solvents such as diethyl ether. It is a strong acid with a weakly coordinating, non-oxidizing conjugate base.[3] It is structurally similar to perchloric acid, but lacks the hazards associated with oxidants.
Structure and production
Pure H[BF4] has not been described. The same holds true for the superacids that are known by the simplified formulas H[PF6] and H[SbF6].[6][7] However, a solution of BF3 in HF is highly acidic, having an approximate speciation of [H2F]+[BF4]− (fluoronium tetrafluoroborate) and a Hammett acidity function of −16.6 at 7 mol % BF3, easily qualifying as a superacid.[8] Although the solvent-free H[BF4] has not been isolated, its solvates are well characterized. These salts consist of protonated solvent as a cation, e.g., H3O+ and H5O+2, and the tetrahedral BF−4 anion. The anion and cations are strongly hydrogen-bonded.[9]
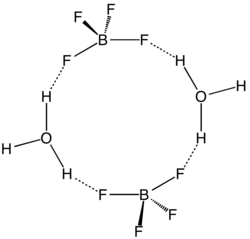
Aqueous solutions of H[BF4] are produced by dissolving boric acid in aqueous hydrofluoric acid.[10][11] Three equivalents of HF react to give the intermediate boron trifluoride and the fourth gives fluoroboric acid:
- B(OH)3 + 4 HF → H3O+ + BF−4 + 2 H2O
An anhydrous fluoroboric acid solution can be prepared by adding aqueous fluoroboric acid to an excess of acetic anhydride at 0 °C (32 °F), which produces a solution of fluoroboric acid, acetic acid, and residual acetic anhydride.[12]
Acidity
Determination of the acidity of fluoroboric acid is complicated by the fact that its name refers to a range of different compounds, e.g. [H(CH3CH2)2O]+[BF4]− (dimethyloxonium tetrafluoroborate), [H3O]+[BF4]− (oxonium tetrafluoroborate), and HF·BF3 (hydrogen fluoride-boron trifluoride 1:1 adduct) – each with a different acidity. The aqueous pKa is quoted as −0.44.[5] Titration of [N((CH2)3CH3)4]+[BF4]− (tetrabutylammonium tetrafluoroborate) in acetonitrile solution indicates that H[BF4], i.e., HF·BF3, has a pKa of 1.6 in that solvent. Its acidity is thus comparable to that of fluorosulfonic acid.[3]
Applications
Fluoroboric acid is the principal precursor to fluoroborate salts, which are typically prepared by treating the metal oxides with fluoroboric acid. The inorganic salts are intermediates in the manufacture of flame-retardant materials and glazing frits, and in electrolytic generation of boron. H[BF4] is also used in aluminum etching and acid pickling.
Organic chemistry
H[BF4] is used as a catalyst for alkylations and polymerizations. In carbohydrate protection reactions, ethereal fluoroboric acid is an efficient and cost-effective catalyst for transacetalation and isopropylidenation reactions. Acetonitrile solutions cleave acetals and some ethers. Many reactive cations have been obtained using fluoroboric acid, e.g. tropylium tetrafluoroborate (C7H7+[BF4]−), triphenylcarbenium tetrafluoroborate (Ph3C]+[BF4]−), triethyloxonium tetrafluoroborate (Et3O]+[BF4]−), and benzenediazonium tetrafluoroborate ([PhN2]+[BF4]−).
Electroplating
Solutions of H[BF4] are used in the electroplating of tin and tin alloys. In this application, methanesulfonic acid is displacing the use of H[BF4].[13] Fluoroboric acid is also used for high-speed electroplating of copper in fluoroborate baths.[14]
Safety
Fluoroboric acid is toxic and attacks skin and eyes. It attacks glass.[5] It hydrolyzes, releasing corrosive, volatile hydrogen fluoride.[13]
Other fluoroboric acids
A series of fluoroboric acids is known in aqueous solutions. The series can be presented as follows:[15]
- H+[B(OH)4]− (hydrogen tetrahydroxyborate) (not a fluoroboric acid)
- H+[BF(OH)3]− (hydrogen fluoro(trihydroxy)borate)
- H+[BF2(OH)2]− (hydrogen difluoro(dihydroxy)borate)
- H+[BF3(OH)]− (hydrogen trifluoro(hydroxy)borate)
- H+[BF4]− (hydrogen tetrafluoroborate)
See also
References
- ^ IUPAC. "Nomenclature of Inorganic Chemistry". Retrieved 2021-04-08.
- ^ a b c Sigma-Aldrich Co., Tetrafluoroboric acid, 48%.
- ^ a b c Kütt, A.; Rodima, T.; Saame, J.; Raamat, E.; Mäemets, V.; Kaljurand, I.; Koppel, I.A.; Garlyauskayte, R.Yu.; Yagupolskii, Y.L.; Yagupolskii, L.M.; Bernhardt, E.; Willner, H.; Leito, I. (21 January 2011). "Equilibrium Acidities of Superacids". The Journal of Organic Chemistry. 76 (2): 391–395. doi:10.1021/jo101409p.
- ^ a b c d "SDS - Tetrafluoroboric acid, 48%". www.thermofisher.com. ThermoFisher Scientific. 27 December 2025. Retrieved 2 February 2026.
- ^ a b c Friestad, Gregory K.; Branchaud, Bruce P. (15 April 2001). "Tetrafluoroboric Acid". Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rt035.
- ^ Juhasz, Mark; Hoffmann, Stephan; Stoyanov, Evgenii; Kim, Kee-Chan; Reed, Christopher A. (2004-10-11). "The Strongest Isolable Acid". Angewandte Chemie International Edition. 43 (40): 5352–5355. doi:10.1002/anie.200460005. ISSN 1433-7851. PMID 15468064.
- ^ Reed, Christopher A. (2005). "Carborane Acids. New "strong yet gentle" acids for organic and inorganic chemistry" (pdf). Chem. Commun. (13): 1669–1677. doi:10.1039/B415425H. ISSN 1359-7345. PMID 15791295.
- ^ Olah, George A.; Surya Prakash, G. K.; Sommer, Jean; Molnar, Arpad (2009-02-03). Superacid chemistry (2nd ed.). Hoboken, N.J.: Wiley. ISBN 9780471596684. OCLC 191809598.
- ^ Mootz, Dietrich; Steffen, Michael (November 1981). "Kristallstrukturen von Säurehydraten und Oxoniumsalzen. XX. Die Oxoniumtetrafluoroborate H
3OBF
4, [H
5O
2]BF
4 und [H(CH
3OH)
2]BF
4" [Crystal structures of acid hydrates and oxonium salts. XX. Oxonium tetrafluoroborates H3OBF4, [H5O2]BF4, and [H(CH3OH)2]BF4]. Zeitschrift für anorganische und allgemeine Chemie (in German). 482 (11): 193–200. doi:10.1002/zaac.19814821124. - ^ Brotherton, R. J.; Weber, C. J.; Guibert, C. R.; Little, J. L. "Boron Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a04_309. ISBN 978-3-527-30673-2.
- ^ Flood, D. T. (1933). "Fluorobenzene". Organic Syntheses. 13: 46. doi:10.15227/orgsyn.013.0046; Collected Volumes, vol. 2, p. 295.
- ^ Wudl, F.; Kaplan, M. L.; Engler, E. M.; Patel, V. V. (January 1979). "2,2′‐Bi‐L,3‐Dithiolylidene (Tetrathiafulvalene, Ttf) and its Radical Cation Salts". Inorganic Syntheses. 19: 27–34. doi:10.1002/9780470132500.ch7.
- ^ a b Balaji, R.; Pushpavanam, Malathy (2003). "Methanesulphonic acid in electroplating related metal finishing industries". Transactions of the Imf. 81 (5): 154–158. doi:10.1080/00202967.2003.11871526. S2CID 91584456.
- ^ Barauskas, Romualdas "Ron" (January 1, 2000). "Copper plating". Metal Finishing. 98 (1): 234–247. doi:10.1016/S0026-0576(00)80330-X. ISSN 0026-0576.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. doi:10.1016/C2009-0-30414-6. ISBN 978-0-08-037941-8.

