Americium(III) fluoride
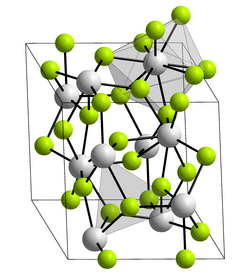 Crystal structure
| |
| Names | |
|---|---|
| IUPAC name
Americium(III) fluoride
| |
| Other names
Americium trifluoride
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
AmF3 |
| Molar mass | 300 g/mol[1] |
| Appearance | pink, crystalline solid |
| Density | 9.53 g/cm3[1] |
| Melting point | 1,393 °C (2,539 °F; 1,666 K)[1] |
| Structure | |
Crystal structure
|
Rhombohedral, hR24 |
Space group
|
P3c1, No. 165[2] |
Lattice constant
|
a = 0.7068 nm, c = 0.7246 nm
|
Lattice volume (V)
|
0.31349 |
Formula units (Z)
|
6 |
| Related compounds | |
Other anions
|
Americium(III) chloride Americium(III) bromide Americium(III) iodide |
Other cations
|
Plutonium(III) fluoride Curium(III) fluoride Europium(III) fluoride |
| Americium(IV) fluoride | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Americium(III) fluoride or americium trifluoride is the chemical compound composed of americium and fluorine with the formula AmF3. It is a water insoluble, pink salt.[3]
References
- ^ a b c Haynes, William M., ed. (2011). CRC Handbook of Chemistry and Physics (92nd ed.). Boca Raton, Florida: CRC Press. p. 4.46. ISBN 1-4398-5511-0.
- ^ Zachariasen, W. H. (1949). "Crystal chemical studies of the 5f-series of elements. XII. New compounds representing known structure types". Acta Crystallographica. 2 (6): 388–390. Bibcode:1949AcCry...2..388Z. doi:10.1107/S0365110X49001016.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 1270–1271. doi:10.1016/C2009-0-30414-6. ISBN 978-0-08-037941-8.