Silver oxalate
 | |
| Names | |
|---|---|
| IUPAC name
Silver(I) ethanedioate
| |
| Other names
Silver Ethanedioate, Silver Salt
Argentous oxalate Silver(I) oxalate | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.007.791 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
Ag 2C 2O 4 |
| Molar mass | 303.755 g/mol |
| Appearance | white powder |
| Density | 5.03 g/cm3 |
| Melting point | 961.9 °C (1,763.4 °F; 1,235.0 K) (decomposes) |
| Boiling point | 2,212 °C (4,014 °F; 2,485 K) at 1013.25 hPa |
Solubility in water
|
3.270*10−3 g/100mL |
Solubility product (Ksp)
|
5.4×10−12[1] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Harmful if swallowed |
| GHS labelling:[2] | |
Pictograms
|
  
|
Signal word
|
Danger |
Hazard statements
|
H201, H302, H312, H410 |
Precautionary statements
|
P210, P230, P240, P250, P264, P270, P273, P280, P301+P317, P302+P352, P317, P321, P330, P362+P364, P370+P380, P372, P373, P391, P401, P501 |
| Safety data sheet (SDS) | External MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
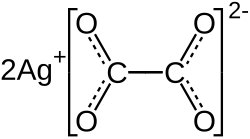
Silver oxalate is a silver salt of oxalic acid with the chemical formula Ag
2C
2O
4. It is sensitive to light, decomposing to metallic silver and carbon dioxide.[3]
Production
Silver oxalate is produced by the reaction between silver nitrate and oxalic acid, or from combining solutions of silver nitrate and sodium oxalate.[3]
Uses
It is commonly employed in experimental petrology to add carbon dioxide (CO
2) to experiments as it will break down to silver (Ag) and carbon dioxide under geologic conditions.[4]
It is also a precursor to the production of silver nanoparticles.
Safety
It is explosive upon heating around 140 °C (284 °F) and the dry material may explode when ground.[5]
See also
- Dioxane tetraketone
References
- ^ John Rumble (June 18, 2018). CRC Handbook of Chemistry and Physics (99 ed.). CRC Press. pp. 5–189. ISBN 978-1138561632.
- ^ PubChem. "Silver oxalate". pubchem.ncbi.nlm.nih.gov. Retrieved 2026-02-26.
- ^ a b E. H. E. Pietsch, A. Kotowski et al. (1975), Gmelin Handbuch der Anorganischen Chemie, Silber Teil B5, 8th edition, p. 148-149, 161
- ^ Silver Oxalate at American Elements
- ^ Silver Oxalate MSDS sheet Archived 2013-12-12 at the Wayback Machine at mpbio