Silver permanganate
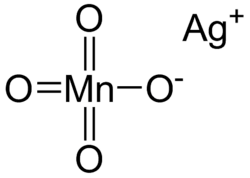 | |
| Names | |
|---|---|
| IUPAC name
Silver(I) permanganate
| |
| Systematic IUPAC name
Silver(I) manganate(VII) | |
| Other names
Argentous permanganate
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.029.127 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
AgMnO4 |
| Molar mass | 226.804 g/mol |
| Appearance | purple crystals or gray powder |
| Density | 4.27 g/cm3 |
| Melting point | 160 °C (320 °F; 433 K) (decomposes) |
Solubility in water
|
0.55 g/100 mL (0 °C) 1.69 g/100 mL (30 °C) |
Magnetic susceptibility (χ)
|
−63.0·10−6 cm3/mol |
| Structure | |
Crystal structure
|
monoclinic |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Eye irritant |
| GHS labelling: | |
Pictograms
|
 
|
Signal word
|
Warning |
Hazard statements
|
H272, H312, H319, H332 |
Precautionary statements
|
P210, P220, P261, P264, P270, P271, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P311, P321, P330, P337+P313, P362+P364, P370+P378, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Silver permanganate is an inorganic compound with the chemical formula AgMnO4. This salt is a purple crystal adopting a monoclinic crystal system.[1] It decomposes when heated or mixed with water, and heating to high temperature may lead to explosion.
Production
It can be produced through the reaction of silver nitrate and potassium permanganate:[2]
- AgNO3 + KMnO4 → AgMnO4 + KNO3
Uses
Silver permanganate is an oxidizing agent for carbon monoxide when deposited on metallic oxide carriers.[3] As a result, it was investigated for use in gas masks but was ultimately abandoned due to its expense and it being consumed in the reaction rather than acting as a catalyst.[4]
Related compounds
A caesium silver permanganate with the formula Cs3Ag[MnO4]4 has been characterized.[5]
References
- ^ Boonstra, E. G. (14 August 1968). "The crystal structure of silver permanganate". Acta Crystallographica Section B. 24 (8): 1053–1062. Bibcode:1968AcCrB..24.1053B. doi:10.1107/S0567740868003699.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. doi:10.1016/C2009-0-30414-6. ISBN 978-0-08-037941-8.
- ^ "The Heterogeneous Oxidation of Carbon Monoxide", Advances in Catalysis, vol. 5, Academic Press, p. 191, 1953-01-01, doi:10.1016/S0360-0564(08)60642-2, retrieved 2026-02-26
- ^ Lamb, Arthur B.; Bray, William C.; Frazer, J. C. W. (1920-03-01). "The Removal of Carbon Monoxide from Air". Journal of Industrial & Engineering Chemistry. 12 (3): 217. doi:10.1021/ie50123a007. ISSN 0095-9014.
- ^ Bauchert, Jörg M.; Henning, Harald; Schleid, Thomas (September 2012). "Synthesis and Crystal Structure of the Cesium Silver Permanganate Cs 3 Ag[MnO 4 ] 4". Zeitschrift für anorganische und allgemeine Chemie (in German). 638 (11): 1780–1783. doi:10.1002/zaac.201200250. ISSN 0044-2313.