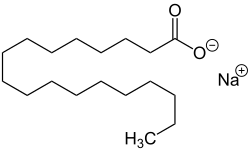
Sodium stearate
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Sodium octadecanoate | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.011.354 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C18H35NaO2 |
| Molar mass | 306.466 g·mol−1 |
| Appearance | white solid |
| Odor | slight, tallow-like odor |
| Density | 1.02 g/cm3 |
| Melting point | 245 to 255 °C (473 to 491 °F; 518 to 528 K) |
Solubility in water
|
soluble |
| Solubility | slightly soluble in ethanediol |
| Hazards | |
| NFPA 704 (fire diamond) | 
2
1
0 |
| Flash point | 176 °C (349 °F; 449 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Sodium stearate (IUPAC: sodium octadecanoate) is the sodium salt of stearic acid. This white solid is the most common soap. It is found in many types of solid deodorants, rubbers, latex paints, and inks. It is also a component of some food additives and food flavorings.[1]
Production
Sodium stearate is produced as a major component of soap upon saponification of oils and fats. The percentage of the sodium stearate depends on the ingredient fats. Tallow is especially high in stearic acid content (as the triglyceride), whereas most fats only contain a few percent. The idealized equation for the formation of sodium stearate from stearin (the triglyceride of stearic acid) follows:[2]
- (C17H35CO2)3C3H5 + 3 NaOH → C3H5(OH)3 + 3 C17H35CO2Na
Sodium stearate can also be made by neutralizing stearic acid with sodium hydroxide.[2]
- C17H35COOH + NaOH → C17H35COONa + H2O
Safety and environmental considerations
Stearate salts, as found in many commercial soaps are of low toxicity, hence their wide use in domestic settings. They do pose some problems for wastewater treatment as they biodegrade relatively slowly and impose a high biological oxygen demand.[1]
References
- ^ a b Schumann, Klaus; Siekmann, Kurt (2000). "Soaps". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a24_247. ISBN 978-3-527-30385-4.
- ^ a b Snell, Foster Dee (1942). "Soap and Glycerol". Journal of Chemical Education. 19 (4): 172. Bibcode:1942JChEd..19..172S. doi:10.1021/ed019p172.